Publications
235. Dey, G.R.; Soliman, S.S.; McCormick, C.R.; Wood, C.H.; Katzbaer, R.R.; Schaak, R.E. “Nanoparticles of Colloidal High Entropy Materials: Capabilities, Challenges, and Opportunities in Synthesis and Characterization,” ACS Nanosci. Au 2024, ASAP Article. 10.1021/acsnanoscienceau.3c00049
234. Alameda, L.T.; Katzbaer, R.R.; Baumler. K.J.; Schaak, R.E. “Soft Chemistry of Hard Materials: Low-Temperature Pathways to Bulk and Nanostructured Layered Metal Borides,” Acc. Chem. Res. 2023, 56, 3515-3524. 10.1021/acs.accounts.3c00579
233. Liz-Marzán, L.M.; Artzi, N.; Bals, S.; Buriak, J.M.; Chan, W.C.W.; Chen, X.; Hersam, M.C.; Kim, I.-D.; Millstone, J.E.; Mulvaney, P.; Parak, W.J.; Rogach, A.; Schaak, R.E. “Celebrating a Nobel Prize to the ‘Discovery of Quantum Dots, an Essential Milestone in Nanoscience’,” ACS Nano 2023, 17, 19474-19475. [Editorial] 10.1021/acsnano.3c09671
232. Hole, B.; Luo, Q.; Garcia, R.; Xie, W.; Rudman, E.; Nguyen, C.L.T.; Dhakal, D.; Young, H.L.; Butterfield, A.G.; Schaak, R.E.; Plass, K.E. “Temperature-Dependent Selection of Reaction Pathways, Reactive Species, and Products During Post-Synthetic Selenization of Copper Sulfide Nanoparticles,” Chem. Mater. 2023, 35, 9073-9085. 10.1021/acs.chemmater.3c01772
231. Veglak, J.M.; Jeong, C.-H.; Young, H.L.; O’Boyle, S.K.; Schaak, R.E. “Using Cation Exchanged Nanorod Templates to Direct the Regioselective Growth and Plasmonic Coupling of Gold Nanoparticles,” ACS Mater. Lett. 2023, 5, 3000-3006. 10.1021/acsmaterialslett.3c01116
230. Young, H.L.; Gomez, E.D.; Schaak, R.E. “Thermally Induced Domain Migration and Interfacial Restructuring in Cation Exchanged ZnS–Cu1.8S Heterostructured Nanorods,” J. Am. Chem. Soc. 2023, 145, 23321-23333. 10.1021/jacs.3c08765
229. Theibault, M.J.; McCormick, C.R.; Schaak, R.E.; Abruña, H.D. “High Entropy Sulfide Nanoparticles as Lithium Polysulfide Redox Catalysts,” ACS Nano 2023, 17, 18402-18410. 10.1021/acsnano.3c05869
228. Schaak, R.E.; Newman, A. “The Artistic Side of ACS Nanoscience Au: Our Cover Art Collection and Tips for Authors,” ACS Nanosci. Au 2023, 3, 266-268. [Editorial] 10.1021/acsnanoscienceau.3c00034
227. He, J.; Alberts, L.; Wang, Y.; Trinquet, V.; Yoshida, S.; Yennawar, H.; Sanni, V.; Claes, R.; Katzbaer, R.; Krysko, E.; Sarker, S.; Schaak, R.E.; Rignanese, G.-M.; Hautier, G.; Mao, Z.; Gopalan, V. “Computationally-driven discovery of second harmonic generation in EuBa3(B3O6)3 through inversion symmetry breaking,” Opt. Mater. Express. 2023, 13, 3416-3427. 10.1364/OME.497727
226. Krysko, E.; Min, L.; Wang, Y.; Zhang, N.; Barber, J.P.; Niculescu, G.E.; Wright, J.; Li, F.; Burrage, K.; Matsuda, M.; Robinson, R.A.; Zhang, Q.; Katzbaer, R. R.; Schaak, R.E.; Terrones, M.; Rost, C.M.; Mao, Z. “Studies on the structure and the magnetic properties of high-entropy spinel oxide (Mg,Mn,Fe,Co,Ni)Al2O4,” APL Materials 2023, 11, 101123. 10.1063/5.0161401
225. Wood, C.R.; Schaak, R.E. “Topochemical Anionic Subunit Insertion Reaction for Constructing Nanoparticles of Layered Oxychalcogenide Intergrowth Structures,” J. Am. Chem. Soc. 2023, 145, 18711-18714. 10.1021/jacs.3c05200
224. Soliman, S.S.; Dey, G.R.; McCormick, C.R.; Schaak, R.E. “Temporal Evolution of Morphology, Composition, and Structure in the Formation of Colloidal High-Entropy Intermetallic Nanoparticles,” ACS Nano 2023, 17, 16147-16159. 10.1021/acsnano.3c05241
223. O’Boyle, S.K.; Baumler, K.J.; Schaak, R.E. “Unexpected Multi-Step Transformation of AgCuS to AgAuS During Nanoparticle Cation Exchange,” Inorg. Chem. 2023, 62, 13050-13057. 10.1021/acs.inorgchem.3c01869
222. Minteer, S.; Brock, S.L.; Masson, G.; Schaak, R.E. “Change is the only constant: A virtual issue on contemporary catalysis,” ACS Measurement Sci. 2023, 3, 237-238. [Editorial] 10.1021/acsmeasuresciau.3c00023
221. McCormick, C.R.; Baksa, S.M.; Veglak, J.M.; Dabo, I.; Schaak, R.E. “Chemical Insights into the Formation of Metastable Zinc Cobalt Sulfide Solid-Solution Nanoparticles through Simultaneous Multi-Cation Exchange,” Chem. Mater. 2023, 35, 5433-5446. 10.1021/acs.chemmater.3c00763
220. Baumler, K.J.; Adams, O.S.; Schaak, R.E. “One-step topochemical transformation of MoAlB into metastable Mo2AlB2 using a metal chloride salt reaction,” Chem. Comm. 2023, 59, 4814-4817. 10.1039/D3CC00138E
219. Katzbaer, R.R.; Vincent, W.M.; Mao, Z.; Schaak, R.E. “Synthesis and Magnetic, Optical, and Electrocatalytic Properties of High Entropy Mixed-Metal Tungsten and Molybdenum Oxides,” Inorg. Chem. 2023, 62, 7843-7852. 10.1021/acs.inorgchem.3c00541
218. Salanne, M.; Buriak, J.M.; Chen, X.; Chueh, W.; Hersam, M.C.; Schaak, R.E. “Best Practices for Simulations and Calculations of Nanomaterials for Energy Applications: Avoiding “Garbage In, Garbage Out,” ACS Nano 2023, 17, 6147-6149. [Editorial] 10.1021/acsnano.3c02368
217. Dey, G.R.; McCormick, C.R.; Soliman, S.S.; Darling, A.J.; Schaak, R.E. “Chemical Insights into the Formation of Colloidal High Entropy Alloy Nanoparticles,” ACS Nano 2023, 17, 5943-5955. 10.1021/acsnano.3c00176
216. Katzbaer, R.R.; Marques dos Santos Vieira, F.; Dabo, I.; Mao, Z.; Schaak, R.E. “Band Gap Narrowing in a High Entropy Spinel Oxide Semiconductor for Enhanced Oxygen Evolution Catalysis,” J. Am. Chem. Soc. 2023, 145, 6753-6761. 10.1021/jacs.2c12887
215. Thompson, K.L.; Katzbaer, R.R.; Terrones, M.; Schaak, R.E. “Formation and Transformation of Cu2-xSe1-yTey Nanoparticles Synthesized by Tellurium Anion Exchange of Copper Selenide,” Inorg. Chem. 2023, 62, 4550-4557. 10.1021/acs.inorgchem.2c04467
214. Buriak, J.M.; Akinwande, D.; Artzi, N.; Brinker, C.J.; Burrows, C.; Chan, W.C.W.; Chen, C.; Chen, X.; Chhowalla, M.; Chi, L.; Chueh, W.; Crudden, C.M. ; Di Carlo, D.; Glotzer, S.C.; Hersam, M.C.; Ho, D.; Hu, T.Y.; Huang, J.; Javey, A.; Kamat, P.V.; Kim, I.-D.; Kotov, N.A.; Lee, T.R.; Lee, Y.H.; Li, Y.; Liz-Marzán, L.M.; Mulvaney, P.; Narang, P.; Nordlander, P.; Oklu, R.; Parak, W.J.; Rogach, A.L.; Salanne, M.; Samorì, P.; Schaak, R.E.; Schanze, K.S.; Sekitani, T.; Skrabalak, S.; Sood, A.K.; Voets, I.K.; Wang, S.; Wang, S.; Wee, A.T.S.; Ye, J. “Best Practices for Using AI When Writing Scientific Manuscripts. Caution, Care, and Consideration: Creative Science Depends on It,” ACS Nano 2023, 17, 4091-4093. [Editorial] 10.1021/acsnano.3c01544
213. Baumler, K.J.; Alameda, L.T.; Katzbaer, R.R.; O’Boyle, S.K.; Lord, R.W.; Schaak, R.E. “Introducing Porosity into Refractory Molybdenum Boride through Controlled Decomposition of a Metastable Mo-Al-B Precursor,” J. Am. Chem. Soc. 2023, 145, 1423-1432. 10.1021/jacs.2c12496
212. McCormick, C.R.; Katzbaer, R.R.; Steimle, B.C.; Schaak, R.E. “Combinatorial Cation Exchange for the Discovery and Rational Synthesis of Heterostructured Nanorods,” Nature Synth. 2023, 2, 152-161. 10.1038/s44160-022-00203-4
211. Young, H.L.; McCormick, C.R.; Butterfield, A.G.; Gomez, E.D.; Schaak, R.E. “Postsynthetic Thiol-Induced Reshaping of Copper Sulfide Nanoparticles,” Chem. Mater. 2022, 34, 11014-11025. 10.1021/acs.chemmater.2c03049
210. Schaak, R.E. “One Year of ACS Nanoscience Au,” ACS Nanosci. Au 2022, 2, 448-449. [Editorial] 10.1021/acsnanoscienceau.2c00056
209. Schaak, R.E. “Increase Your Impact: Writing Tips to Reach a Broader Audience,” ACS Nanoscience Au 2022, 2, 250-252. [Editorial] 10.1021/acsnanoscienceau.2c00030
208. Katzbaer, R.R.; Theibault, M.J.; Kirchner-Hall, N.E.; Mao, Z.; Dabo, I.; Abruña, H.D.; Schaak, R.E. “Understanding the Photoelectrochemical Properties of Theoretically Predicted Water-Splitting Catalysts for Effective Materials Discovery,” Adv. Energy Mater. 2022, 2201869. 10.1002/aenm.202201869
207. Dong, X.; Katzbaer, R.R.; Chitara, B.; Yang, L.; Schaak, R.E.; Yan, F. “Optimizing the synergistic effect of CuWO4–CuS hybrid composites for photocatalytic inactivation of pathogenic bacteria,” Environ. Sci. Nano. 2022, 9, 4283-4294.
206. Kotov, N.A.; Akinwande, D.; Brinker, C.J.; Buriak, J.M.; Chan, W.C.W.; Chen, X.; Chhowalla, M.; Chueh, W.; Glotzer, S.C.; Gogotsi, Y.; Hersam, M.C.; Ho, D.; Hu, T.; Javey, A.; Kagan, C.R.; Kataoka, K.; Kim, I.-D.; Lee, S.-T.; Lee, Y.H.; Liz-Marzan, L.M.; Millstone, J.E.; Mulvaney, P.; Nel, A.E.; Nordlander, P.; Parak, W.J.; Penner, R.M.; Rogach, A.L.; Salanne, M.; Schaak, R.E.; Sood, A.K.; Stevens, M.; Tsukruk, V.; Wee, A.T.S.; Voets, I.; Weil, T.; Weiss, P.S. “Tanks and Truth,” ACS Nano 2022, 16, 4975-4976. [Editorial] 10.1021/acsnano.2c02602
205. Schaak, R.E. “Designer Nanostructrues in ACS Nanoscience Au,” ACS Nanoscience Au 2022, 2, 1-2. [Editorial] 10.1021/acsnanoscienceau.2c00003
204. Wang, H.; Katzbaer, R.R.; Fanghanel, J.; Schaak, R.E.; Gopalan, V. “Ultrasensitive electrode-free and co-catalyst-free detection of nano moles per hour hydrogen evolution for the discovery of new photocatalysts,” Rev. Sci. Inst. 2022, 93, 025002. 10.1063/5.0077650
203. O’Boyle, S.K.; Fagan, A.M.; Steimle, B.C.; Schaak, R.E. “Expanded Tunability of Intraparticle Frameworks in Spherical Heterostructured Nanoparticles through Substoichiometric Partial Cation Exchange,” ACS Materials Au 2022, 2, 690-698. 10.1021/acsmaterialsau.2c00038
202. Jeffries, W.R.; Fagan, A.M.; Schaak, R.E.; Knappenberger Jr., K.L. “Influence of Band Alignment on Electronic Relaxation in Plasmonic Metal-Semiconductor Hybrid Nanoparticles,” J. Phys. Chem. C 2022, 126, 8384-8392. 10.1021/acs.jpcc.2c01378
201. Fagan, A.M.; Steimle, B.C.; Schaak, R.E. “Orthogonal reactivity and interface-driven selectivity during cation exchange of heterostructured metal sulfide nanorods,” Chem. Comm. 2022, 58, 4328-4331. 10.1039/D1CC07190D
200. Katzbaer, R.R.; Zhu, Y.; Mao, Z.; Schaak, R.E. “Persistence and Evolution of Materials Features During Catalysis Using Topological and Trivial Polymorphs of MoTe2,” ChemCatChem 2022, 14, e202101714. 10.1002/cctc.202101714
199. Li, Z.; Kang, L.; Lord, R.W.; Park, K.; Gillman, A.; Vaia, R.A.; Schaak, R.E.; Werner, D.H.; Knappenberger Jr., K.L. “Plasmon-mediated Chiroptical Second Harmonic Generation from Seemingly Achiral Gold Nanorods,” ACS Nanoscience Au 2022, 2, 32-39. 10.1021/acsnanoscienceau.1c00014
198. Millstone, J.E.; Eikey, E.A.; Fagan, A.M.; Gan, X.Y.; Lord, R.W.; Sen, R.; Steimle, B.C.; Schaak, R.E. “Virtual Issue on Nanosynthetic Chemistry,” ACS Nano 2021, 15, 13893-13896. [Editorial] 10.1021/acsnano.1c08046
197. Seth, K.; Darling, A.J.; Holder, C.F.; Xiong, Y.; Shallenberger, J.R.; Schaak, R.E. “In Situ Evolution of Ru4Al13 Crystals into a Highly Active Catalyst for the Hydrogen Evolution Reaction,” Chem. Mater. 2021, 33, 7124-7131. 10.1021/acs.chemmater.1c02583
196. Butterfield, A.G.; McCormick, C.R.; Veglak, J.M.; Schaak, R.E. “Morphology-Dependent Phase Selectivity of Cobalt Sulfide during Nanoparticle Cation Exchange Reactions,” J. Am. Chem. Soc. 2021, 143, 7915-7919. 10.1021/jacs.1c03478
195. Garcia-Herrera, L.F.; McAllister, H.P.; Xiong, H.; Wang, H.; Lord, R.W.; O’Boyle, S.K.; Imamovic, A.; Steimle, B.C.; Schaak, R.E.; Plass, K.E. “Multi-Step Regioselectivity and Non-Kirkendall Anion Exchange of Copper Chalcogenide Nanorods,” Chem. Mater. 2021, 33, 3841-3850. 10.1021/acs.chemmater.1c01107
194. Di Domizio, G.A.; Alameda, L.T.; Fanghanel, J.; Lord, R.W.; Miller. J.R.; Schaak, R.E. “Real-Time Monitoring of Competing Nanoparticle Formation Pathways during Cation Exchange Using Benchtop Light Scattering,” Chem. Mater. 2021, 33, 3936-3944. 10.1021/acs.chemmater.0c04938
193. Xiong, Y.; Campbell, Q.T.; Fanghanel, J.; Badding, C.K.; Wang, H.; Kirchner- Hall, N.E.; Theibault, M.J.; Timrov, I.; Mondschein, J.S.; Seth, K.; Katz, R.; Molina Villarino, A.; Pamuk, B.; Penrod, M.E.; Khan, M.M.; Rivera, T.; Smith, N.C.; Quintana, X.; Orbe, P.; Fennie, C.J.; Asem-Hiablie, S.; Young, J.L.; Deutsch, T.G.; Cococcioni, M.; Gopalan, V.; Abruña, H.D.; Schaak, R.E.; Dabo, I. “Optimizing accuracy and efficacy in data-driven materials discovery for the solar production of hydrogen,” Energy Environ. Sci. 2021, 14, 2335-2348. 10.1039/D0EE02984J
192. Sanchez-Cano, C.; Alvarez-Puebla, R.A.; Abendroth, J.M.; Beck, T.; Blick, R.; Cao, Y.; Caruso, F.; Chakraborty, I.; Chapman, H.N.; Chen, C.; Cohen, B.E.; Conceição, A.L.C.; Cormode, D.P.; Cui, D.; Dawson, K.A.; Falkenberg, G.; Fan, C.; Feliu, N.; Gao, M.; Gargioni, E.; Glüer, C.-C.; Grüner, F.; Hassan, M.; Hu, Y.; Huang, Y.; Huber, S.; Huse, N.; Kang, Y.; Khademhosseini, A.; Keller, T.F.; Körnig, C.; Kotov, N.A.; Koziej, D.; Liang, X.-J.; Liu, B.; Liu, S.; Liu, Y.; Liu, Z.; Liz-Marzán, L.M.; Ma, X.; Machicote, A.; Maison, W.; Mancuso, A.P.; Megahed, S.; Nickel, B.; Otto, F.; Palencia, C.; Pascarelli, S.; Pearson, A.; Peñate-Medina, O.; Qi, B.; Rädler, J.; Richardson, J.J.; Rosenhahn, A.; Rothkamm, K.; Rübhausen, M.; Sanyal, M.K.; Schaak, R.E.; Schlemmer, H.-P.; Schmidt, N.; Schmutzler, O.; Schotten, T.; Schulz, F.; Sood, A.K.; Spiers, K.M.; Staufer, T.; Stemer, D.M.; Stierle, A.; Sun, X.; Tsakanova, G.; Weiss, P.S.; Weller, H.; Westermeier, F.; Xu, N.; Yan, H.; Zeng, Y.; Zhao, Y.; Zhao, Y.; Zhu, D.; Zhu, Y.; Parak, W.J. “X-ray-Based Techniques to Study the Nano–Bio Interface,” ACS Nano 2021, 15, 3754-3807. 10.1021/acsnano.0c09563
191. Cheon, J.; Manna, L.; Schaak, R.E. “Why do we care about studying transformations in inorganic nanocrystals?” Acc. Chem. Res. 2021, 54, 1543-1544. [Editorial] 10.1021/acs.accounts.1c00123
190. Hernández-Pagán, E.; Lord, R.W.; Veglak, J.M.; Schaak, R.E. “Incorporation of Metal Phosphide Domains into Colloidal Hybrid Nanoparticles,” Inorg. Chem. 2021, 60, 4278-4290. 10.1021/acs.inorgchem.0c03826
189. Sun, Y.; Terrones, M.T.; Schaak, R.E. “Colloidal Nanostructures of Transition Metal Dichalcogenides,” Acc. Chem. Res. 2021, 54, 1517-1527. 10.1021/acs.accounts.1c00006
188. Butterfield, A.G.; Alameda, L.T.; Schaak, R.E. “Emergence and Control of Stacking Fault Formation during Nanoparticle Cation Exchange Reactions,” J. Am. Chem. Soc. 2021, 143, 1779–1783. 10.1021/jacs.0c13072
187. McCormick, C.R.; Schaak, R.E. “Simultaneous Multication Exchange Pathway to High-Entropy Metal Sulfide Nanoparticles,” J. Am. Chem. Soc. 2021, 143, 1017-1023. 10.1021/jacs.0c11384
186. Fagan, A.M.; Jeffries, W.R.; Knappenberger Jr, K.L.; Schaak, R.E. “Synthetic Control of Hot-Electron Thermalization Efficiency in Size-Tunable Au–Pt Hybrid Nanoparticles,” ACS Nano 2021, 15, 1378-1387. 10.1021/acsnano.0c08661
185. Brinker, C.J.; Buriak, J.M.; Chan, W.C.W.; Chhowalla, M.; Glotzer, S.C.; Gogotsi, Y.; Hammond, P.T.; Hersam, M.C.; Javey, A.; Kagan, C.R.; Kataoka, K.; Khademhosseini, A.; Kim, I.-D.; Kotov, N.A.; Lee, S.-T.; Lee, Y.H.; Li, Y.; Liz-Marzán, L.M.; Millstone, J.M.; Mulvaney, P.; Nel, A.E.; Nordlander, P.; Parak, W.J.; Penner, R.M.; Rogach, A.L.; Schaak, R.E.; Sood, A.K.; Stevens, M.M.; Wee, A.T.S.; Weil, T.; Wilson, C.G.; Weiss, P.S. “Growing Contributions of Nano in 2020,” ACS Nano 2020, 14, 16163-16164. [Editorial] 10.1021/acsnano.0c10429
184. Lord, R.W.; Fanghanel, J.; Holder, C.F.; Dabo, I.; Schaak, R.E. “Colloidal Nanoparticles of a Metastable Copper Selenide Phase with Near-Infrared Plasmon Resonance,” Chem. Mater. 2020, 32, 10227-10234. 10.1021/acs.chemmater.0c04058
183. Schaak, R.E.; Steimle, B.C.; Fenton, J.L. “Made-to-Order Heterostructured Nanoparticle Libraries,” Acc. Chem. Res. 2020, 53, 2558-2568. 10.1021/acs.accounts.0c00520
182. Schaak, R.E.; Penner, R.M.; Buriak, J.M.; Caruso, F.M.; Chhowalla, M.; Gogotsi, Y.; Mulvaney, P.; Parak, W.J.; Weiss, P.S. “Tutorials and Articles on Best Practices,” ACS Nano 2020, 14, 10751-10753. [Editorial] 10.1021/acsnano.0c07588
181. Butterfield, A.G.; Steimle, B.C.; Schaak, R.E. “Retrosynthetic Design of Morphologically Complex Metal Sulfide Nanoparticles Using Sequential Partial Cation Exchange and Chemical Etching,” ACS Materials Lett. 2020, 2, 1106-1114. 10.1021/acsmaterialslett.0c00287
180. Steimle, B.C.; Lord, R.W.; Schaak, R.E. “Phosphine-Induced Phase Transition in Copper Sulfide Nanoparticles Prior to Initiation of a Cation Exchange Reactions,” J. Am. Chem. Soc. 2020, 142, 13345-13349. 10.1021/jacs.0c06602
179. Holder, C.F.; Fanghanel, J.; Xiong, Y.; Dabo, I.; Schaak, R.E. “Phase-Selective Solution Synthesis of Perovskite-Related Cesium Cadmium Chloride Nanoparticles,” Inorg. Chem. 2020, 59, 11688-11694. 10.1021/acs.inorgchem.0c01574
178. Darling, A.J.; Stewart, S.; Holder, C.F.; Schaak, R.E. “Bulk-immiscible AgRh Alloy Nanoparticles as a Highly Active Electrocatalyst for the Hydrogen Evolution Reaction,” ChemNanoMat 2020, 6, 1320-1324. 10.1002/cnma.202000381
177. Steimle, B.C.; Fagan, A.M.; Butterfield, A.G.; Lord, R.W.; McCormick, C.R.; Di Domizio, G.A.; Schaak, R.E. “Experimental Insights into Partial Cation Exchange Reactions for Synthesizing Heterostructured Metal Sulfide Nanocrystals,” Chem. Mater. 2020, 32, 5461-5482. 10.1021/acs.chemmater.0c01388
176. Schaak, R.E. “Beyond Guano-Doped Graphene,” ACS Nano 2020, 14, 2555-2556. [Editorial] 10.1021/acsnano.0c02181
175. Sun, Y.; Wang, Y.; Chen, J.Y.C.; Fujisawa, K.; Holder, C.F.; Miller, J.T.; Crespi, V.H.; Terrones, M.; Schaak, R.E. “Interface-mediated noble metal deposition on transition metal dichalcogenide nanostructures,” Nature Chem. 2020, 12, 284-293. 10.1038/s41557-020-0418-3
174. Steimle, B.C.; Fenton, J.L.; Schaak, R.E. “Rational construction of a scalable heterostructured nanorod megalibrary,” Science 2020, 367, 418-424. 10.1126/science.aaz1172
173. Sun, Y.; Darling, A.J.; Li, Y.; Fujisawa, K.; Holder, C.F.; Liu, H.; Janik, M.J.; Terrones, M.; Schaak, R.E. “Defect-Mediated Selective Hydrogenation of Nitroarenes on Nanostructured WS2,” Chem. Sci. 2019, 10, 10310-10317. 10.1039/C9SC03337H
172. Holder, C.F.; Schaak, R.E. “Tutorial on Powder X-Ray Diffraction for Characterizing Nanoscale Materials,” ACS Nano 2019, 13, 7359-7365. 10.1021/acsnano.9b05157
171. Fenton, J.L.; Fagan, A.M.; Schaak, R.E. “General Solution-Phase Synthesis of Nanoscale Transition Metal Tellurides Using Metal Nanoparticle Reagents,” Eur. J. Inorg. Chem. 2019, 2019, 3490-3493. 10.1002/ejic.201900560
170. Alameda, L.T.; Lord, R.W.; Barr, J.A.; Moradifar, P.; Metzger, Z.P.; Steimle, B.C.; Holder, C.F.; Alem, N.; Sinnott, S.B.; Schaak, R.E. “Multi-Step Topochemical Pathway to Metastable Mo2AlB2 and Related Two-Dimensional Nanosheet Heterostructures,” J. Am. Chem. Soc. 2019, 141, 10852-10861. 10.1021/jacs.9b04726
169. Millstone, J.E.; Chan, W.C.W.; Kagan, C.R.; Liz-Marzán, L.M.; Kotov, N.A.; Mulvaney, P.A.; Parak, W.J.; Rogach, A.L.; Weiss, P.S.; Schaak, R.E. “Redefining the Experimental and Methods Sections,” ACS Nano 2019, 13, 4862-4864. [Editorial] 10.1021/acsnano.9b03753
168. Lord, R.W.; Holder, C.F.; Fenton, J.L.; Schaak, R.E. “Seeded Growth of Metal Nitrides on Noble-Metal Nanoparticles to Form Complex Nanoscale Heterostructures,” Chem. Mater. 2019, 31, 4605-4613. 10.1021/acs.chemmater.9b01638
167. Li, X.; Schaak, R.E. “ZnO-Templated Synthesis and Metal-Insulator Transition of VO2 Nanostructures,” Chem. Mater. 2019, 31, 2088-2096. 10.1021/acs.chemmater.8b05231
166. Fenton, J.L.; Steimle, B.C.; Schaak, R.E. “Structure-Selective Synthesis of Wurtzite and Zincblende ZnS, CdS, and CuInS2 Using Nanoparticle Cation Exchange Reactions,” Inorg. Chem. 2019, 58, 672-678. 10.1021/acs.inorgchem.8b02880
165. Chan, W.C.W.; Chhowalla, M.; Farokhzad, O.; Glotzer, S.; Gogotsi, Y.; Hammond, P.T.; Hersam, M.C.; Javey, A.; Kagan, C.R.; Kataoka, K.; Khademhosseini, A.; Kotov, N.A.; Lee, S.-T.; Lee, Y.H.; Li, Y.; Millstone, J.E.; Mulvaney, P.; Nel, A.E.; Nordlander, P.J.; Parak, W.J.; Penner, R.M.; Rogach, A.L.; Schaak, R.E.; Sood, A.K.; Stevens, M.M.; Wee, A.T.S.; Weil, T.; Willson, C.G.; Weiss, P.S. “The 15th Anniversary of the U.S. National Nanotechnology Initiative,” ACS Nano 2018, 12, 10567-10569. [Editorial] 10.1021/acsnano.8b08676
164. Smith, W.C.; Morse, J.R.; Bria, C.R.M.; Schaak, R.E.; Williams, S.K.R. “Composition-Based Separation of Pt-Fe3O4 Hybrid Nanoparticles by Thermal Field-Flow Fractionation,” ACS Appl. Nano Mater. 2018, 1, 6435-6443. 10.1021/acsanm.8b01643
163. Voiry, D.; Chhowalla, M.; Gogotsi, Y.; Kotov, N.A.; Li, Y.; Penner, R.M.; Schaak, R.E.; Weiss, P.S. “Best Practices for Reporting Electrocatalytic Performance of Nanomaterials,” ACS Nano 2018, 12, 9635-9638. [Editorial] 10.1021/acsnano.8b07700
162. Alameda, L.T.; Moradifar, P.; Metzger, Z.P.; Alem, N.; Schaak, R.E. “Topochemical Deintercalation of Al from MoAlB: Stepwise Etching Pathway, Layered Intergrowth Structures, and Two-Dimensional MBene,” J. Am. Chem. Soc. 2018, 140, 8833-8840. 10.1021/jacs.8b04705
161. Fenton, J.L.; Steimle, B.C.; Schaak, R.E. “Exploiting Crystallographic Regioselectivity to Engineer Asymmetric Three-Component Colloidal Nanoparticle Isomers using Partial Cation Exchange Reactions,” J. Am. Chem. Soc. 2018, 140, 6771-6775. 10.1021/jacs.8b03338
160. Fenton, J.L.; Steimle, B.C.; Schaak, R.E. “Tunable Intraparticle Frameworks for Creating Complex Heterostructured Nanoparticle Libraries,” Science 2018, 360, 513-517. 10.1126/science.aar5597
159. Mondschein, J.S.; Kumar, K.; Holder, C.F.; Seth, K.; Kim, H.; Schaak, R.E. “Intermetallic Ni2Ta Electrocatalyst for the Oxygen Evolution Reaction in Highly Acidic Electrolytes,” Inorg. Chem. 2018, 57, 6010-6015. 10.1021/acs.inorgchem.8b00503
158. Badding, C.K.; Soucy, T.L.; Mondschein, J.S.; Schaak, R.E. “Metal Ruthenate Perovskites as Heterogeneous Catalysts for the Hydrolysis of Ammonia Borane,” ACS Omega 2018, 3, 3501-3506. 10.1021/acsomega.7b02003
157. Parak, W.J.; Chan, W.C.W.; Chhowalla, M.; Farokhzad, O.; Glotzer, S.; Gogotsi, Y.; Hammond, P.T.; Hersam, M.C.; Javey, A.; Kagan, C.R.; Kataoka, K.; Khademhosseini, A.; Kotov, N.A.; Lee, S.-T. Lee, Y.H.; Li, Y.; Millstone, J.; Mulvaney, P.A.; Nel, A.E.; Nordlander, P.J.; Penner, R.M.; Rogach, A.L.; Schaak, R.E.; Stevens, M.M.; Wee, A.T.S.; Willson, C.G.; Weiss, P.S. “Helmuth Möhwald (1946-2018),” ACS Nano 2018, 12, 3053-3055. [Editorial] 10.1021/acsnano.8b02755
156. Schaak, R.E. “Author Profile: Raymond Schaak,” Angew. Chem. Int. Ed. 2018, 57, 3002. 10.1002/anie.201711024
155. Chan, W.C.W.; Chhowalla, M.; Farokhzad, O.; Glotzer, S.; Gogotsi, Y.; Hafner, J.H.; Hammond, P.T.; Hersam, M.C.; Javey, A.; Kagan, C.R.; Kataoka, K.; Khademhosseini, A.; Kotov, N.A.; Lee, S.-T.; Li, Y.; Millstone, J.; Möhwald, H.; Mulvaney, P.C.; Nel, A.E.; Nordlander, P.J.; Parak, W.J.; Penner, R.M.; Rogach, A.L.; Schaak, R.E.; Stevens, M.M.; Wee, A.T.S.; Willson, C.G.; Fernandez, L.E.; Weiss, P.S. “A Big Year Ahead for Nano in 2018,” ACS Nano 2017, 11, 11755-11757. 10.1021/acsnano.7b08851
154. Alameda, L.T.; Holder, C.F.; Fenton, J.L.; Schaak, R.E. “Partial Etching of Al from MoAlB Single Crystals to Expose Catalytically-Active Basal Planes for the Hydrogen Evolution Reaction,” Chem. Mater. 2017, 29, 8953-8957. 10.1021/acs.chemmater.7b02511
153. Li, X.; Schaak, R.E. “Size and Interface Modulated Metal-Insulator Transition in Solution-Synthesized Nanoscale VO2-TiO2-VO2 Heterostructures,” Angew. Chem. Int. Ed. 2017, 56, 15550-15554.
152. Sun, Y.; Fujisawa, K.; Terrones, M.; Schaak, R.E. “Solution synthesis of few-layer WTe2 and MoxW1-xTe2 nanostructures,” J. Mater. Chem. C 2017, 5, 11317-11323. 10.1039/C7TC02860A
151. Ledendecker, M.; Mondschein, J.S.; Kasian, O.; Geiger, S.; Göhl, D.; Schalenbach, M.; Zeradjanin, A.; Cherevko, S.; Schaak, R.E.; Mayrhofer, K. “Stability and Activity of Non-Noble-Metal-Based Catalysts Toward the Hydrogen Evolution Reaction,” Angew. Chem. Int. Ed. 2017, 56, 9767-9771.
150. Sun, Y.; Fujisawa, K.; Lin, Z.; Lei, Y.; Mondschein, J.S.; Terrones, M.; Schaak, R.E. “Low-Temperature Solution Synthesis of Transition Metal Dichalcogenide Alloys with Tunable Optical Properties,” J. Am. Chem. Soc. 2017, 139, 11096-11105. 10.1021/jacs.7b04443
149. Chan, W.C.W.; Chhowalla, M.; Glotzer, S.; Gogotsi, Y.; Hafner, J.H.; Hammond, P.T.; Hersam, M.C.; Javey, A.; Kagan, C.R.; Kataoka, K.; Khademhosseini, A.; Kotov, N.A.; Lee, S.T.; Li, Y.; Möhwald, H.; Mulvaney, P.; Nel, A.E.; Nordlander, P.J.; Parak, W.J.; Penner, R.M.; Rogach, A.L.; Schaak, R.E.; Stevens, M.M.; Wee, A.T.S.; Willson, C.G.; Fernandez, L.E.; Weiss, P.S. “Our First and Next Decades at ACS Nano,” ACS Nano 2017, 11, 7553-7555. [Editorial] 10.1021/acsnano.7b05765
148. Fenton, J.L.; Hodges, J.M.; Schaak, R.E. “Synthetic Deconvolution of Interfaces and Materials Components in Hybrid Nanoparticles,” Chem. Mater. 2017, 29, 6168-6177. 10.1021/acs.chemmater.7b02556
147. Hodges, J.M.; Schaak, R.E. “Controlling Configurational Isomerism in Three-Component Colloidal Hybrid Nanoparticles,” Acc. Chem. Res. 2017, 50, 1433-1440. 10.1021/acs.accounts.7b00105
146. Li, X.; Schaak, R.E. “Reactive AgAuS and Ag3AuS2 Synthons Enable the Sequential Transformation of Spherical Nanocrystals into Asymmetric Multi-Component Hybrid Nanoparticles,” Chem. Mater. 2017, 29, 4153-4160. 10.1021/acs.chemmater.7b01449
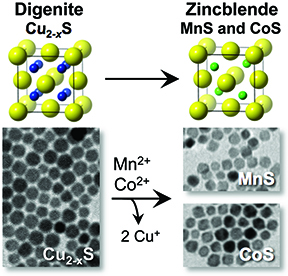
145. Fenton, J.L.; Schaak, R.E. “Structure-Selective Cation Exchange in the Synthesis of Zincblende MnS and CoS Nanocrystals,” Angew. Chem. Int. Ed. 2017, 56, 6464-6467. 10.1002/anie.201701087
144. Morse, J.R.; Callejas, J.F.; Darling, A.J.; Schaak, R.E. “Bulk Iron Pyrite as a Catalyst for the Selective Hydrogenation of Nitroarenes,” Chem. Commun. 2017, 53, 4807-4810. 10.1039/C7CC00120G
143. Bria, C.R.M.; Skelly, P.W.; Morse, J.R.; Schaak, R.E.; Ratanathanawongs Williams, S.K. “Semi-Preparative Asymmetrical Flow Field-Flow Fractionation: A Closer Look at Channel Dimensions and Separation Performance,” J. Chromatogr. A 2017, 1499, 149-157. 10.1016/j.chroma.2017.03.017
142. Pelaz, B.; Alexiou, C; Alvarez Puebla, R.A.; Alves, F.; Andrews, A.M.; Ashraf, S.; Balogh, L.P.; Ballerini, L.; Bestetti, A.; Brendel, C.; Bosi, S.; Carril, M.; Chan, W.C.W.; Chen, C.; Chen, S.; Chen, X.; Cheng, Z.; Cui, D.; Du, J.; Dullin, C.; Escudero, A.; Feliu, N.; Gao, M.; George, M.; Grünweller, A.; Gu, Z.; Gogotsi, Y.; Halas, N.J.; Hampp, N.; Hartmann, R.K.; Hersam, M.C.; Hunziker, P.; Jian, J.; Jiang, X.; Jungebluth, P.; Kadhiresan, P.; Kataoka, K.; Khademhosseini, A.; Kopecek, J.; Kotov, N.A.; Krug, H.F.; Lee, D.S.; Lehr, C.-M.; Leong, K.W.; Liang, X.-J.; Lim, M.; Liz Marzán, L.M.; Ma, X.; Macchiarini, P.; Meng, H.; Möhwald, H.; Mulvaney, P.; Nel, A.E.; Nie, S.; Nordlander, P.; Okano, T.; Oliveira, J.; Park, T.H.; Penner, R.M.; Prato, M.; Puntes, V.; Rotello, V.; Samarakoon, A.; Schaak, R.E.; Shen, Y.; Sjoqvist, S.; Skirtach, A.G.; Soliman, M.G.; Stevens, M.M.; Tang, B.Z.; Tietze, R.; VanEpps, S.; Udugama, B.N.; Sung, H.-W.; Weil, T.; Weiss, P.S.; Willner, I.; Wu, Y.; Yang, L.; Yue, Z.; Zhang, Q.; Zhang, Q.; Zhang, X.-E.; Zhao, Y.; Zhou, X.; Parak, W.J. “Diverse Applications of Nanomedicine,” ACS Nano 2017, 11, 2313-2381. 10.1021/acsnano.6b06040
141. Khademhosseini, A.; Chan, W.C.W.; Chhowalla, M.; Glotzer, S.C.; Gogotsi, Y.; Hafner, J.H.; Hammond. P.T.; Hersam, M.C.; Javey, A.; Kagan, C.R.; Kotov, N.A.; Lee, S.-T.; Li, Y.; Möhwald, M.; Mulvaney, P.A.; Nel, A.E.; Parak, W.J.; Penner, R.M.; Rogach, A.L.; Schaak, R.E.; Stevens, .M.; Wee, A.T.S.; Brinker, J.; Chen, X.; Chi, L.; Crommie, M.; Dekker, C.; Farokhzad, O.; Gerber, C.; Ginger, D.S.; Irvine, D.J.; Kiessling, L.L.; Kostarelos, K.; Landes, C.; Lee, T.; Leggett, G.J.; Liang, X.-J.; Marzan, L.L.; Millstone, J.; Odom, T.W.; Ozcan, A.; Prato, M.; Rao, C.N.R.; Sailor, M.J.; Weiss, E.; Weiss, P.S. “Nanoscience and Nanotechnology Cross Borders,” ACS Nano 2017, 11, 1123-1126. [Editorial] 10.1021/acsnano.7b00953
140. Mondschein, J.S.; Callejas, J.F.; Read, C.G.; Chen, J.Y.C.; Holder, C.F.; Badding, C.K.; Schaak, R.E. “Crystalline Cobalt Oxide Films for Sustained Electrocatalytic Oxygen Evolution under Strongly Acidic Conditions,” Chem. Mater. 2017, 29, 950-957. 10.1021/acs.chemmater.6b02879
139. Sun, D.S.; Xiong, Y.; Sun, Y.; Dabo, I.; Schaak, R.E. “Solution-Synthesized In4SnSe4 Semiconductor Microwires with a Direct Band Gap,” Chem. Mater. 2017, 29, 1095-1098. 10.1021/acs.chemmater.6b04216
138. Sun, D.S.; Schaak, R.E. “Solution-Mediated Growth of Two-Dimensional SnSe@GeSe Nanosheet Heterostructures,” Chem. Mater. 2017, 29, 817-822. 10.1021/acs.chemmater.6b04808
137. Hodges, J.M.; Morse, J.R.; Fenton, J.L.; Ackerman, J.D.; Alameda, L.T.; Schaak, R.E. “Insights into the Seeded-Growth Synthesis of Colloidal Hybrid Nanoparticles,” Chem. Mater. 2017, 29, 106-119. 10.1021/acs.chemmater.6b02795
136. Lin, Z.; McCreary, A.; Briggs, N.; Subramanian, S.; Zhang, K.H.; Sun, Y.F.; Li, X.F.; Borys, N.J.; Yua, H.T.; Fullerton-Shirey, S.K.; Chernikov, A.; Zhao, H.; McDonnell, S.; Lindenberg, A.M.; Xiao, K.; LeRoy, B.J.; Drndic, M.; Hwang, J.C.M.; Park, J.; Chhowalla, M.; Schaak, R.E.; Javey, A.; Hersam, M.C.; Robinson, J.; Terrones, M. “2D materials advances: from large scale synthesis and controlled heterostructures to improved characterization techniques, defects and applications,” 2D Materials 2016, 3, 042001. 10.1088/2053-1583/3/4/042001
135. Chan, W.W.C.; Chhowalla, M.; Glotzer, S.; Gogotsi, Y.; Hafner, J.H.; Hammond, P.T.; Hersam, M.C.; Javey, A.; Kagan, C.R.; Khademhosseini, A.; Kotov, N.A.; Lee, S.T.; Li, Y.; Mohwald, H.; Mulvaney, P.A.; Nel, A.E.; Nordlander, P.J.; Parak, W.J.; Penner, R.M.; Rogach, A.L.; Schaak, R.E.; Stevens, M.M.; Wee, A.T.S.; Willson, C.G.; Fernandez, L.E.; Weiss, P.S. “Nanoscience and Nanotechnology Impacting Diverse Fields of Science, Engineering, and Medicine,” ACS Nano 2016, 10, 10615-10617. [Editorial] 10.1021/acsnano.6b08335
134. J.F. Callejas, C.G. Read, C.W. Roske, N.S. Lewis, R.E. Schaak, “Synthesis, Characterization, and Properties of Metal Phosphide Catalysts for the Hydrogen-Evolution Reaction,” Chem. Mater. 2016, 28, 6017-6044. 10.1021/acs.chemmater.6b02148
133. C.G. Read, J.F. Callejas, C.F. Holder, R.E. Schaak, “General Strategy for the Synthesis of Transition Metal Phosphide Films for Electrocatalytic Hydrogen and Oxygen Evolution,” ACS Appl. Mater. Interfaces 2016, 8, 12798-12803. 10.1021/acsami.6b02352
132. A.E. Powell, J.M. Hodges, R.E. Schaak, “Preserving Both Anion and Cation Sublattice Features during a Nanocrystal Cation Exchange Reaction: Synthesis of Metastable Wurtzite-Type CoS and MnS,” J. Am. Chem. Soc. 2016, 138, 471-474. 10.1021/jacs.5b10624
131. Y. Sun, Y. Wang, D. Sun, B.R. Carvalho, C.G. Read, C.-H. Lee, Z. Lin, K. Fujisawa, J.A. Robinson, V.H. Crespi, M. Terrones, R.E. Schaak, “Low-Temperature Solution Synthesis of Few-Layer 1T’-MoTe2 Nanostructures Exhibiting Lattice Compression,” Angew. Chem. Int. Ed. 2016, 55, 2830-2834. 10.1002/anie.201510029
130. A.M. Wiltrout, C.G. Read, E.M. Spencer, R.E. Schaak, “Solution Synthesis of Thiospinel CuCo2S4 Nanoparticles,” Inorg. Chem. 2016, 55, 221-226. 10.1021/acs.inorgchem.5b02158
129. J.M. McEnaney, T.L. Soucy, J.M. Hodges, J.F. Callejas, J.S. Mondschein, R.E. Schaak, “Colloidally-Synthesized Cobalt Molybdenum Nanoparticles as Active and Stable Electrocatalysts for the Hydrogen Evolution Reaction under Alkaline Conditions,” J. Mater. Chem. A 2016, 4, 3077-3081. 10.1039/C5TA07055D
128. W.W.C. Chan, S. Glotzer, Y. Gogotsi, J.H. Hafner, P.T. Hammond, M.C. Hersam, A. Javey, C.R. Kagan, A. Khademhosseini, N.A. Kotov, S.T. Lee, H. Mohwald, P.A. Mulvaney, A.E. Nel, P.J. Nordlander, W.J. Parak, R.M. Penner, A.L. Rogach, R.E. Schaak, M.M. Stevens, A.T.S. Wee, C.G. Willson, H.L. Tierney, P.S. Weiss, “Grand Plans for Nano,” ACS Nano 2015, 9, 11503-11505. [Editorial] 10.1021/acsnano.5b07280
127. G.R. Bhimanapati, Z. Lin, V. Meunier, Y. Jung, J. Cha, S. Das, D. Xiao, Y. Son, M.S. Strano, V.R. Cooper, L. Liang, S.G. Louie, E. Ringe, W. Zhou, S.S. Kim, R.R. Naik, B.G. Sumpter, H. Terrones, F. Xia, Y. Wang, J. Zhu, D. Akinwande, N. Alem, J.A. Schuller, R.E. Schaak, M. Terrones, J.A. Robinson, “Recent Advances in Two-Dimensional Materials Beyond Graphene,” ACS Nano 2015, 9, 11509-11539. 10.1021/acsnano.5b05556
126. J.M. Hodges, J.R. Morse, M.E. Williams, R.E. Schaak, “Microscopic Investigation of Chemoselectivity in Ag-Pt-Fe3O4 Heterotrimer Formation: Mechanistic Insights and Implications for Controlling High-Order Hybrid Nanoparticle Morphology,” J. Am. Chem. Soc. 2015, 137, 15493-15500. 10.1021/jacs.5b10254
125. C.G. Read, T.R. Gordon, J.M. Hodges, R.E. Schaak, “Colloidal Hybrid Nanoparticle Insertion Reaction for Transforming Heterodimers into Heterotrimers,” J. Am. Chem. Soc. 2015, 137, 12517-12517. DOI: 10.1021/jacs.5b08850
124. J.M. Hodges, J.L. Fenton, J.L. Gray, R.E. Schaak, “Colloidal ZnO and Zn1-xCoxO Tetrapod Nanocrystals with Tunable Arm Lengths,” Nanoscale 2015, 7, 16671-16676. DOI: 10.1039/c5nr04425a
123. J.M. Hodges, K. Kletetschka, J.L. Fenton, C.G. Read, R.E. Schaak, “Sequential Anion and Cation Exchange Reactions for Complete Material Transformations of Nanoparticles with Morphological Retention,” Angew. Chem. Int. Ed. 2015, 54, 8669-8672. [Designated as a “Hot Paper” by the editors of Angew. Chem. Int. Ed.; featured on the front cover of Angew. Chem. Int. Ed.] DOI: 10.1002/anie.201504099
122. D. Sun, S. Feng, M. Terrones, R.E. Schaak, “Reversible Laser-Modulated Interlayer Decoupling in Colloidal MoSe2 Nanoflowers,” Chem. Mater. 2015, 27, 3167-3175. DOI: 10.1021/acs.chemmater.5b01129
121. J.F. Callejas, C.G. Read, E.J. Popczun, R.E. Schaak, “Nanostructured Co2P Electrocatalyst for the Hydrogen Evolution Reaction and Direct Comparison with Morphologically Equivalent CoP,” Chem. Mater. 2015, 27, 3769-3774. DOI: 10.1021/acs.chemmater.5b01284
120. M.J. Bradley, C.G. Read, R.E. Schaak, “Pt-Au Nanoparticle Heterodimers as Seeds for Pt-Au-Metal Sulfide Heterotrimers: Thermal Stability and Chemoselective Growth Characteristics,” J. Phys. Chem. C. 2015, 119, 8952-8959. DOI: 10.1021/acs.jpcc.5b01274
119. C.W. Roske, E.J. Popczun, B.S. Seger, C.G. Read, T. Pedersen, O. Hansen, P.C.K. Vesborg, B.S. Brunschwig, R.E. Schaak, I. Chorkendorff, H.B. Gray, N.S. Lewis, “Comparison of the performance of CoP-coated and Pt-coated radial junction n+p-silicon microwire-array photocathodes for the sunlight-driven reduction of water to H2(g),” J. Phys. Chem. Lett. 2015, 6, 1679-1683. DOI: 10.1021/acs.jpclett.5b00495
118. R.E. Schaak, “Appealing to Our Broad Audience,” ACS Nano 2015, 9, 1005-1007. [Editorial] DOI: 10.1021/acsnano.5b00961
117. A.J. Biacchi, R.E. Schaak, “Ligand-Induced Fate of Embryonic Seeds in the Shape-Controlled Synthesis of Rhodium Nanoparticles,” ACS Nano 2015, 9, 1707-1720. DOI: 10.1021/nn506517e
116. E.J. Popczun, C.W. Roske, C.G. Read, J.C. Crompton, J.M. McEnaney, J.F. Callejas, N.S. Lewis, R.E. Schaak, “Highly branched cobalt phosphide nanostructures for hydrogen-evolution electrocatalysis,” J. Mater. Chem. A 2015, 3, 5420-5425. DOI: 10.1039/C4TA06642A
115. R. Lv, J.A. Robinson, R.E. Schaak, D. Sun, Y. Sun, T.E. Mallouk, M. Terrones, “Transition Metal Dichalcogenides and Beyond: Synthesis, Properties, and Applications of Single- and Few-Layer Nanosheets,” Acc. Chem. Res. 2015, 48, 56-64. DOI: 10.1021/ar5002846
114. J.M. McEnaney, R.E. Schaak, “Solution Synthesis of Metal Silicide Nanoparticles,” Inorg. Chem. 2015, 54, 707-709. DOI: 10.1021/ic502394u
113. Chan, W. C. W., Gogotsi, Y.; Hafner, J. H.; Mohwald, H.; Hammond, P. T.; Mulvaney, P. A.; Hersam, M. C.; Nel, A. E.; Javey, A.; Nordlander, P. J.; Kagan, C. R.; Parak, W. J.; Khademhosseini, A.; Penner, R. M.; Jotov, N. A.; Rogach, A. L.; Lee, S. T.; Schaak, R. E.; Stevens, M. M.; Willson, C. G.; Wee, A. T. S.; Weiss, P. S. “A Year for Nanoscience,” ACS Nano 2014, 8, 11901-11903. [Editorial] DOI: 10.1021/nn5070716
112. D.D. Vaughn II, J. Araujo, P. Meduri, J.F. Callejas, M.A. Hickner, R.E. Schaak, “Solution Synthesis of Cu3PdN Nanocrystals as Ternary Metal Nitride Electrocatalysts for the Oxygen Reduction Reaction,” Chem. Mater. 2014, 26, 6226-6232. DOI: 10.1021/cm5029723
111. T.R. Gordon, R.E. Schaak, “Synthesis of Hybrid Au-In2O3 Nanoparticles Exhibiting Dual Plasmonic Resonance,” Chem. Mater. 2014, 26, 5900-5904. [Featured on the cover of Chemistry of Materials, October 28 2014 issue.] DOI: 10.1021/cm502396d
110. J.F. Callejas, J.M. McEnaney, C.G. Read, J.C. Crompton, A.J. Biacchi, E.J. Popczun, T.R. Gordon, N.S. Lewis, R.E. Schaak, “Electrocatalytic and Photocatalytic Hydrogen Production from Acidic and Neutral-pH Aqueous Solutions Using Iron Phosphide Nanoparticles,” ACS Nano 2014, 8, 11101-11107. [Featured in In Nano, December 2014 issue of ACS Nano.] DOI: 10.1021/nn5048553
109. J.M. McEnaney, J.C. Crompton, J.F. Callejas, E.J. Popczun, C.G. Read, N.S. Lewis, R.E. Schaak, “Electrocatalytic Hydrogen Evolution Using Amorphous Tungsten Phosphide Nanoparticles,” Chem. Comm. 2014, 50, 11026-11028. DOI: 10.1039/C4CC04709E
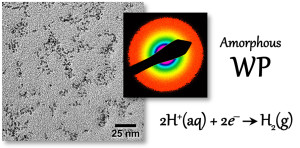
108. J.M. McEnaney, J.C. Crompton, J.F. Callejas, E.J. Popczun, A.J. Biacchi, N.S. Lewis, R.E. Schaak, “Amorphous Molybdenum Phosphide Nanoparticles for Electrocatalytic Hydrogen Evolution,” Chem. Mater. 2014, 26, 4826-4831. DOI: 10.1021/cm502035s
107. E.J. Popczun, C.G. Read, C.W. Roske, N.S. Lewis, R.E. Schaak, “Highly active electrocatalysis of the hydrogen evolution reaction by cobalt phosphide nanoparticles,” Angew. Chem. Int. Ed. 2014, 53, 5427-5430. [Designated as a “hot paper” by the editors of Angew. Chem. Int. Ed.] DOI: 10.1002/anie.201402646
106. M.R. Buck, A.J. Biacchi, R.E. Schaak, “Insights into the Thermal Decomposition of Co(II) Oleate for the Shape-Controlled Synthesis of Wurtzite-Type CoO Nanocrystals,” Chem. Mater. 2014, 26, 1492-1499. DOI: 10.1021/cm4041055
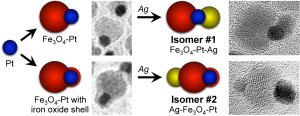
105. J.M. Hodges, A.J. Biacchi, R.E. Schaak, “Ternary Hybrid Nanoparticle Isomers: Directing the Nucleation of Ag on Pt-Fe3O4 Using a Solid-State Protecting Group,” ACS Nano 2014, 8, 1047-1055. DOI: 10.1021/nn405943z
104. W.C.W. Chan, Y. Gogotsi, J.H. Hafner, P.T. Hammond, M.C. Hersam, A. Javey, C.R. Kagan, A. Khademhosseini, N.A. Kotov, H. Mohwald, P.A. Mulvaney, A.E. Nel, P.J. Nordlander, W.J. Parak, R.M. Penner, A.L. Rogach, R.E. Schaak, M.M. Stevens, A.T.S. Wee, C.G. Willson, P.S. Weiss, “Exciting Times for Nano,” ACS Nano 2013, 7, 10437-10439. [Editorial] DOI: 10.1021/nn4064055
103. C.G. Read, A.J. Biacchi, R.E. Schaak, “Au-Ge and Ag-Ge Heterodimers with Tunable Domain Sizes: A Supersaturation-Precipitation Route to Colloidal Hybrid Nanoparticles,” Chem. Mater. 2013, 25, 4304-4311. DOI: 10.1021/cm4024452
102. D.D. Vaughn II, D. Sun, J.A. Moyer, A.J. Biacchi, R. Misra, P. Schiffer, R.E. Schaak, “Solution-Phase Synthesis and Magnetic Properties of Single-Crystal Iron Germanide Nanostructures,” Chem. Mater. 2013, 25, 4396-4401. DOI: 10.1021/cm402795r
101. A.J. Biacchi, D.D. Vaughn II, R.E. Schaak, “Synthesis and Crystallographic Analysis of Shape-Controlled SnS Nanocrystal Photocatalysts: Evidence for a Pseudotetragonal Structural Modification,” J. Am. Chem. Soc. 2013, 135, 11634-11644. DOI: 10.1021/ja405203e
100. E.J. Popczun, J.R. McKone, C.G. Read, A.J. Biacchi, A.M. Wiltrout, N.S. Lewis, R.E. Schaak, “Nanostructured Nickel Phosphide as an Electrocatalyst for the Hydrogen Evolution Reaction,” J. Am. Chem. Soc. 2013, 135, 9267-9270. DOI: 10.1021/ja403440e
99. E.R. Essinger-Hileman, E.J. Popczun, R.E. Schaak, “Magnetic separation of colloidal nanoparticle mixtures using a material specific peptide,” Chem. Comm. 2013, 49, 5471-5473. DOI: 10.1039/C3CC42496K
98. M.R. Buck, A.J. Biacchi, E.J. Popczun, R.E. Schaak, “Polymer-Assisted Synthesis of Colloidal Germanium Telluride Nano-Octahedra, Nanospheres, and Nanosheets,” Chem. Mater. 2013, 25, 2163-2171. DOI: 10.1021/cm4009656
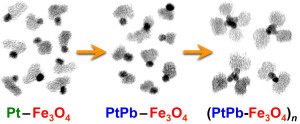
97. M.J. Bradley, A.J. Biacchi, R.E. Schaak, “Chemical Transformation of Pt-Fe3O4 Colloidal Hybrid Nanoparticles into PtPb-Fe3O4 and Pt3Sn-Fe3O4 Heterodimers and (PtPb-Fe3O4)n Nanoflowers,” Chem. Mater. 2013, 25, 1886-1892. [Featured on the cover of the July 23, 2013 issue of Chem. Mater.] DOI: 10.1021/cm4005163
96. M.R. Buck and R.E. Schaak, “Emerging Strategies for the Total Synthesis of Inorganic Nanostructures,” Angew. Chem. Int. Ed. 2013, 52, 6154-6178. DOI: 10.1002/anie.201207240
95. D.D. Vaughn II and R.E. Schaak, “Synthesis, properties and applications of colloidal germanium and germanium-based nanomaterials,” Chem. Soc. Rev. 2013, 42, 2861-2879. DOI: 10.1039/C2CS35364D
94. D.A. Bonnell, J.M. Buriak, W.C.W. Chan, J.H. Hafner, P.T. Hammond, M.C. Hersam, A. Javey, N.A. Kotov, A.E. Nel, P.J. Norlander, W. Parak, R.M. Penner, A.L. Rogach, R.E. Schaak, M.M. Stevens, A.T.S. Wee, C.G. Willson, P.S. Weiss, “We Take It Personally,” ACS Nano 2012, 6, 10417-10419. [Editorial] DOI: 10.1021/nn305696y
93. R.E. Schaak and M.E. Williams, “Full Disclosure: The Practical Side of Nanoscale Total Synthesis,” ACS Nano 2012, 6, 8492-8497. [Perspective] DOI: 10.1021/nn304375v
92. D.D. Vaughn II, D. Sun, S.M. Levin, A.J. Biacchi, T.S. Mayer, and R.E. Schaak, “Colloidal Synthesis and Electrical Properties of GeSe Nanobelts,” Chem. Mater. 2012, 24, 3643-3649. [Featured on the cover of the Jan 8, 2013 issue of Chem. Mater.] DOI: 10.1021/cm3023192
91. I.T. Sines, D.D. Vaughn II, E.J. Popczun, and R.E. Schaak, “Synthesis of Tetragonal Mackinawite-Type FeS Nanosheets by Solvothermal Crystallization,” J. Solid State Chem. 2012, 196, 17-20. DOI: 10.1016/j.jssc.2012.07.056
90. I.T. Sines, D.D. Vaughn II, A.J. Biacchi, C.E. Kingsley, E.J. Popczun, and R.E. Schaak, “Engineering Porosity into Single-Crystal Colloidal Nanosheets Using Epitaxial Nucleation and Chalcogenide Anion Exchange Reactions: The Conversion of SnSe to SnTe,” Chem. Mater. 2012, 24, 3088-3093. DOI: 10.1021/cm301734b
89. J.M. Buriak, R.E. Schaak, and P.S. Weiss, “In Response,” ACS Nano 2012, 6, 3643-3645. [Editorial] DOI: 10.1021/nn3019046
88. D.D. Vaughn II, O.D. Hentz, S. Chen, D. Wang, and R.E. Schaak, “Formation of SnS Nanoflowers for Lithium Ion Batteries,” Chem. Comm. 2012, 48, 5608-5610. DOI: 10.1039/C2CC32033A
87. N.E. Motl, J.F. Bondi, and R.E. Schaak, “Synthesis of Colloidal Au-Cu2S Heterodimers via Chemically Triggered Phase Segregation of AuCu Nanoparticles,” Chem. Mater. 2012, 24, 1552-1554. DOI: 10.1021/cm300511q
86. D.A. Bonnell, J.M. Buriak, J.H. Hafner, P.T. Hammond, M.C. Hersam, A. Javey, N.A. Kotov, P. Nordlander, W.J. Parak, A.L. Rogach, R.E. Schaak, M.M. Stevens, A.T.S. Wee, C.G. Willson, P.S. Weiss, “Recycling is Not Always Good: The Dangers of Self-Plagiarism,” ACS Nano 2012, 6, 1-4. [Editorial] DOi: 10.1021/nn3000912
85. S.-I. In, D.D. Vaughn II, and R.E. Schaak, “Hybrid CuO-TiO2-xNx Hollow Nanocubes for Photocatalytic CO2 Conversion to Methane under Solar Irradiation,” Angew. Chem. Int. Ed. 2012, 51, 3915-3918. [Designated as a “hot paper” by the editors of Angew. Chem. Int. Ed.] DOI: 10.1002/anie.201108936
84. M.R. Buck, J.F. Bondi, and R.E. Schaak, “A total synthesis framework for the construction of high-order colloidal hybrid nanoparticles,” Nature Chemistry 2012, 4, 37-44. [Featured as “Editor’s Choice” in Science and highlighted in Chemical & Engineering News, RSC’s Chemistry World, and MRS Bulletin] DOI: 10.1038/nchem.1195
83. H.L. Tierney, D.A. Bonnell, J.M. Buriak, J.H. Hafner, P.T. Hammond, A.L. Rogach, M.C. Hersam, R.E. Schaak, A. Javey, M.M. Stevens, N.A. Kotov, A.T.S. Wee, P. Nordlander, C.G. Willson, W.J. Parak, P.S. Weiss, “ACS Nano in 2011 and Looking Forward to 2012,” ACS Nano 2011, 5, 9301-9302. [Editorial] DOI: 10.1021/nn204760v
82. D.D. Vaughn II and R.E. Schaak, “A Precursor-Limited Nanoparticle Coalescence Pathway for Tuning the Thickness of Laterally-Uniform Colloidal Nanosheets: The Case of SnSe,” ACS Nano 2011, 5, 8852-8860. DOI: 10.1021/nn203009v
81. R.E. Schaak, “Comprehensive and Full,” ACS Nano 2011, 5, 6767-6768. [Editorial] DOI: 10.1021/nn203388d
80. A.J. Biacchi and R.E. Schaak, “The Solvent Matters: Kinetic vs. Thermodynamic Shape Control in the Polyol Synthesis of Rhodium Nanoparticles,” ACS Nano 2011, 5, 8089-8099. DOI: 10.1021/nn2026758
79. J.S. Beveridge, M.R. Buck, J.F. Bondi, R. Misa, P. Schiffer, R.E. Schaak, and M.E. Williams, “Purification and Magnetic Interrogation of Hybrid Au-Fe3O4 and FePt-Fe3O4 Nanoparticles,” Angew. Chem. Int. Ed. 2011, 50, 9875-9879. [Designated by Angew. Chem. Int. Ed. as a “Very Important Paper” and featured in Chemical & Engineering News and Science Daily] DOI: 10.1002/anie.201104829
78. J.F. Bondi and R.E. Schaak, “Solution Chemistry Synthesis of Intermetallic Gold-Lithium Nanoparticles,” Eur. J. Inorg. Chem. 2011, 3877-3880. [Featured on the cover of Eur. J. Inorg. Chem. and highlighted by ACS Noteworthy Chemistry] DOI: 10.1002/ejic.201100276
77. Z.L. Schaefer, K.M. Weeber, R. Misra, P. Schiffer, and R.E. Schaak, “Bridging hcp-Ni and Ni3C via a Ni3C1-x Solid Solution: Tunable Composition and Magnetism in Colloidal Nickel Carbide Nanoparticles,” Chem. Mater. 2011, 23, 2475-2480. DOI: 10.1021/cm200410
76. Z. Luo; Y. Vasquez; J.F. Bondi; and R.E. Schaak, “Pawley and Rietveld refinements using electron diffraction from L12-type intermetallic Au3Fe1-x nanocrystals during their in-situ order-disorder transition,” Ultramicroscopy 2011, 111, 1295-1304. DOI: 10.1016/j.ultramic.2011.04.003
75. E.R. Essinger-Hileman, D. DeCicco, J.F. Bondi, and R.E. Schaak, “Aqueous room-temperature synthesis of Au-Rh, Au-Pt, Pt-Rh, and Pd-Rh alloy nanoparticles: Fully tunable compositions within the miscibility gaps,” J. Mater. Chem. 2011, 21, 11599-11604. DOI: 10.1039/C0JM03913F
74. I.T. Sines and R.E. Schaak, “Phase-Selective Chemical Extraction of Selenium and Sulfur from Nanoscale Metal Chalcogenides: A General Strategy for Synthesis, Purification, and Phase Targeting,” J. Am. Chem. Soc. 2011, 133, 1294-1297. DOI: 10.1021/ja110374d
73. W.J. Parak, P.S. Weiss, D.A. Bonnell, J.M. Buriak, J.H. Hafner, P.T. Hammond, M.C. Hersam, N.A. Kotov, R.E. Schaak, C.G. Willson, “Virtual Issue on Nanotoxicology,” ACS Nano 2010, 4, 5513-5514. [Editorial] DOI: 10.1021/nn1026406
72. D.D. Vaughn II and R.E. Schaak, “Single Crystal Colloidal Nanosheets of GeS and GeSe,” J. Am. Chem. Soc. 2010, 132, 15170-15172. DOI: 10.1021/ja107520b
71. N. Motl, E. Ewusi-Annan, I.T. Sines, L. Jensen, and R.E. Schaak, “Au-Cu Alloy Nanoparticles with Tunable Compositions and Plasmonic Properties: Experimental Determination of Composition and Correlation with Theory,” J. Phys. Chem. C 2010, 114, 19263-19269. DOI: 10.1021/jp107637j
70. D.D. Vaughn II and R.E. Schaak, “Colloidal Synthesis of Air-Stable Crystalline Germanium Nanoparticles with Tunable Sizes and Shapes,” Chem. Mater. 2010, 22, 6103-6108. DOI: 10.1021/cm1015965
69. Z.L. Schaefer, M.L. Gross, M.A. Hickner, and R.E. Schaak, “Uniform Hollow Carbon Shells: Nanostructured Graphitic Supports for Improved Oxygen-Reduction Catalysis,” Angew. Chem. Int. Ed. 2010, 49, 7045-7048. [Designated as a “hot paper” by the editors of Angew. Chem. Int. Ed.] DOI: 10.1002/anie.201003213
68. M.E. Anderson, S.S.N. Bharadwaja, and R.E. Schaak, “Modified Polyol Synthesis of Bulk-Scale Nanostructured Bismuth Antimony Telluride,” J. Mater. Chem. 2010, 20, 8362-8367. DOI: 10.1039/C0JM01424A
67. J.F. Bondi, R. Misra, X. Ke, I.T. Sines, P. Schiffer, and R.E. Schaak, “Optimized Synthesis and Magnetic Properties of Intermetallic Au3Fe1-x, Au3Ni1-x, and Au3Co1-x Nanoparticles,” Chem. Mater. 2010, 22, 3988-3994. DOI: 10.1021/cm100705c
66. M.R. Buck and R.E. Schaak, “Liquid-Phase Synthesis of Uniform Cube-Shaped GeTe Microcrystals,” Chem. Mater. 2010, 22, 3236-3240. DOI: 10.1021/cm1004483
65. I.T. Sines, R. Misra, P. Schiffer, and R.E. Schaak, “Colloidal Synthesis of Non-Equilibrium Wurtzite-Type MnSe,” Angew. Chem. Int. Ed. 2010, 49, 4638-4640. [Featured on the inside cover of the 27th issue of Angew. Chem. Int. Ed., June 21, 2010] DOI: 10.1002/anie.201001213, 10.1002/anie.201003242
64. N.L. Henderson, X. Ke, P. Schiffer, and R.E. Schaak, “Solution Precursor Synthesis and Magnetic Properties of Eu1-xCaxTiO3,” J. Solid State Chem. 2010, 183, 631-635. DOI: 10.1016/j.jssc.2009.12.025
63. Z.L. Schaefer, D.D. Vaughn, and R.E. Schaak, “Solution Chemistry Synthesis, Morphology Studies, and Optical Properties of Five Distinct Nanocrystalline Au-Zn Intermetallic Compounds,” J. Alloys Compounds, 2010, 490, 98-102. DOI: 10.1016/j.jallcom.2009.10.137
62. N.H. Chou, K.D. Oyler, N.E. Motl, and R.E. Schaak, “Colloidal Synthesis of Germanium Nanocrystals Using Room-Temperature Benchtop Chemistry,” Chem. Mater. 2009, 21, 4105-4107. DOI: 10.1021/cm902088y
61. Q. Liu, K.K. Gath, J.C. Bauer, R.E. Schaak, and J.H. Lunsford, “The active phase in the direct synthesis of H2O2 from H2 and O2 over Pd/SiO2 catalyst in a H2SO4/ethanol system,” Catal. Lett. 2009, 132, 342-348. DOI: 10.1007/s10562-009-0104-y
60. K.D. Oyler, X. Ke, I.T. Sines, P.E. Schiffer, and R.E. Schaak, “Chemical Synthesis of Two-Dimensional Iron Chalcogenide Nanosheets: FeSe, FeTe, Fe(Se,Te), and FeTe2,” Chem. Mater. 2009, 21, 3655-3661. DOI: 10.1021/cm901150c
59. J.F. Bondi, K.D. Oyler, X. Ke, P.E. Schiffer, and R.E. Schaak, “Chemical Synthesis of Air-Stable Manganese Nanoparticles,” J. Am. Chem. Soc. 2009, 131, 9144-9145. DOI: 10.1021/ja901372q
58. K.N. Avery, J.E. Schaak, and R.E. Schaak, “M13 Bacteriophage as a Biological Scaffold for Magnetically-Recoverable Metal Nanowire Catalysts: Combining Specific and Non-Specific Interactions to Design Multi-Functional Nanocomposites,” Chem. Mater. 2009, 21, 2176-2178. [Featured in Chemical & Engineering News, June 1, 2009] DOI: 10.1021/cm900869u
57. T.H. Phan and R.E. Schaak, “Polyol Synthesis of Palladium Hydride: Bulk Powders vs. Nanocrystals,” Chem. Commun. 2009, 3026-3029. DOI: 10.1039/B902024A
56. B.M. Leonard, M.E. Anderson, K.D. Oyler, T.H. Phan, and R.E. Schaak, “Orthogonal Reactivity of Metal and Multi-Metal Nanostructures for Selective, Stepwise, and Spatially-Controlled Solid State Modification,” ACS Nano 2009, 3, 940-948. [Highlighted in a Perspective article by Catherine Murphy, pg. 770-774 (same issue)] DOI: 10.1021/nn800892a
55. Q. Liu, Z. Yan, J.C. Bauer, N.L. Henderson, D.W. Goodman, J.D. Batteas, and R.E. Schaak, “Synthesis of CuPt Nanorod Catalysts with Tunable Lengths,” J. Am. Chem. Soc. 2009, 131, 5720-5721. DOI: 10.1021/ja810151r
54. N.L. Henderson, M.D. Straesser, P.D. Sabato, and R.E. Schaak, “Toward Green Metallurgy: Low-Temperature Solution Synthesis of Bulk-Scale Intermetallic Compounds in Edible Plant and Seed Oils,” Green Chem. 2009, 11, 974-978. DOI: 10.1039/B815443K
53. F. Dawood and R.E. Schaak, “ZnO-Templated Synthesis of Wurtzite-Type ZnS and ZnSe Nanoparticles,” J. Am. Chem. Soc. 2009, 131, 424-425. DOI: 10.1021/ja808455u, 10.1021/ja9058316
52. Z.L. Schaefer, X. Ke, P.E. Schiffer, and R.E. Schaak, “Direct Solution Synthesis, Reaction Pathway Studies, and Structural Characterization of Crystalline Ni3B Nanoparticles,” J. Phys. Chem. C 2008, 112, 19846-19851. DOI: 10.1021/jp8082503
51. Y. Vasquez, Z. Luo, and R.E. Schaak, “Low-Temperature Solution Synthesis of the Non-Equilibrium Ordered Intermetallic Compounds Au3Fe, Au3Co, and Au3Ni as Nanocrystals,” J. Am. Chem. Soc. 2008, 130, 11866-11867. DOI: 10.1021/ja804858u
50. M.E. Anderson, M.R. Buck, I.A. Sines, K.D. Oyler, and R.E. Schaak, “On-Wire Conversion Chemistry: Engineering Solid State Complexity into Striped Metal Nanowires using Solution Chemistry Reactions,” J. Am. Chem. Soc. 2008, 130, 14042-14043. DOI: 10.1021/ja804743b
49. A.E. Henkes and R.E. Schaak, “Synthesis of Nanocrystalline REBO3 (RE = Y, Nd, Sm, Eu, Gd, Ho) and YBO3:Eu Using a Borohydride-Based Solution Precursor Route,” J. Solid State Chem. 2008, 181, 3264-3268. DOI: 10.1016/j.jssc.2008.08.028
48. R.E. Schaak, “Solid State Chemistry on the Nanoscale: Achievements, Challenges, and Opportunities,” J. Solid State Chem. 2008, 181, 1507-1508. [Editorial] DOI: 10.1016/j.jssc.2008.06.035
47. Y. Vasquez, A.E. Henkes, J.C. Bauer, and R.E. Schaak, “Nanocrystal Conversion Chemistry: A Unified and Materials-General Strategy for the Template Based Synthesis of Nanocrystalline Solids,” J. Solid State Chem. 2008, 181, 1509-1523. DOI: 10.1016/j.jssc.2008.04.007
46. Q. Liu, J.C. Bauer, R.E. Schaak, and J.H. Lunsford, “Supported Palladium Nanoparticles: An Efficient Catalyst for the Direct Formation of H2O2 from H2 and O2,” Angew. Chem. Int. Ed. 2008, 47, 6221-6224. DOI: 10.1002/anie.200801517
45. N.H. Chou, X. Ke, P.E. Schiffer, and R.E. Schaak, “Room-Temperature Chemical Synthesis of Shape-Controlled Indium Nanoparticles,” J. Am. Chem. Soc. 2008, 130, 8140-8141. DOI: 10.1021/ja801949c
44. N.L. Henderson and R.E. Schaak, “Low Temperature Solution-Mediated Synthesis of Polycrystalline Intermetallic Compounds from Bulk Metal Powders,” Chem. Mater. 2008, 20, 3212-3217. DOI: 10.1021/cm800245j
43. N.H. Chou and R.E. Schaak, “A Library of Single Crystal Metal-Tin Nanorods: Using Diffusion as a Tool for Controlling the Morphology of Intermetallic Nanocrystals,” Chem. Mater. 2008, 20, 2081-2085. DOI: 10.1021/cm703640u
42. Q. Liu, J.C. Bauer, R.E. Schaak, and J.H. Lunsford, “Direct Synthesis of H2O2 from H2 and O2 over PdPt/SiO2 Bimetallic Catalyst in a H2SO4/Ethanol System,” Appl. Catal. A 2008, 339, 130-136. DOI: 10.1016/j.apcata.2008.01.026
41. J.C. Bauer, X. Chen, Q. Liu, T.-H. Phan, and R.E. Schaak, “Converting Nanocrystalline Metals into Alloys and Intermetallic Compounds for Applications in Catalysis,” J. Mater. Chem. 2008, 18, 275-282. [Featured as the cover article for the 3rd issue of J. Mater. Chem., January 2008; Featured in Science (January 18, 2008, “Editor’s Choice”)] DOI: 10.1039/B712035D
40. A.E. Henkes and R.E. Schaak, “Template Assisted Synthesis of Shape-Controlled Rh2P Nanocrystals,” Inorg. Chem. 2008, 47, 671-677. DOI: 10.1021/ic701783f
39. F.A. Dawood, B.M. Leonard, and R.E. Schaak, “Oxidative Transformation of Intermetallic Nanoparticles: An Alternative Pathway to Metal/Oxide Nanocomposites, Textured Ceramics, and Multi-Metal Oxides,” Chem. Mater. 2007, 19, 4545-4550. DOI: 10.1021/cm071147t
38. R.E. Cable and R.E. Schaak, “Solution Synthesis of Nanocrystalline M-Zn (M = Pd, Au, Cu) Intermetallic Compounds via Chemical Conversion of Metal Nanoparticle Precursors,” Chem. Mater. 2007, 19, 4098-4104. DOI: 10.1021/cm071214j
37. A.E. Henkes and R.E. Schaak, “Trioctylphosphine: A General Phosphorus Source for the Low-Temperature Conversion of Metals into Transition Metal Phosphides,” Chem. Mater. 2007, 19, 4234-4242. DOI: 10.1021/cm071021w
36. N.H. Chou and R.E. Schaak, “Shape-Controlled Conversion of b-Sn Nanocrystals into Intermetallic M-Sn (M = Fe, Co, Ni, Pd) Nanocrystals,” J. Am. Chem. Soc. 2007, 129, 7339-7345. DOI: 10.1021/ja069032y
35. N. Henderson, J. Baek, P.S. Halasyamani, and R.E. Schaak, “Ambient Pressure Synthesis of SHG-Active Eu2Ti2O7 with a [110] Layered Perovskite Structure: Suppressing Pyrochlore Formation by Oxidation of Perovskite-Type EuTiO3,” Chem. Mater. 2007, 19, 1883-1885. DOI: 10.1021/cm062934d, 10.1021/cm7029807
34. A.E. Henkes, Y. Vasquez, and R.E. Schaak, “Converting Metals into Phosphides: A General Strategy for the Synthesis of Metal Phosphide Nanocrystals,” J. Am. Chem. Soc. 2007, 129, 1896-1897. DOI: 10.1021/ja068502l
33. R.E. Schaak, “Structure and Chemistry of Crystalline Solids. By Bodie E. Douglas and Shih-Ming Ho (University of Pittsburgh). Springer Science + Business Media, Inc.: New York. 2006. x + 346 pp. $129.00. ISBN: 0-387-26147-8,” J. Am. Chem. Soc. 2007, 129, 451-452. [Invited book review] DOI: 10.1021/ja069778w
32. R.E. Cable and R.E. Schaak, “Reacting the Unreactive: A Toolbox of Low-Temperature Solution-Mediated Reactions for the Facile Interconversion of Nanocrystalline Intermetallic Compounds,” J. Am. Chem. Soc. 2006, 128, 9588-9589. [Featured in Nature (July 27, 2006, “Research Highlights”)] DOI: 10.1021/ja0627996
31. B.M. Leonard and R.E. Schaak, “Multistep Solution-Mediated Formation of Nanocrystalline AuCuSn2: Mechanistic Insights for the Guided Design of Intermetallic Solid State Materials and Complex Multi-Metal Nanocrystals,” J. Am. Chem. Soc. 2006, 128, 11475-11482. DOI: 10.1021/ja062475h
30. A.K. Sra, T.D. Ewers, Q. Xu, H. Zandbergen, and R.E. Schaak, “One-Pot Synthesis of Bi-Disperse FePt Nanoparticles and Spontaneous Assembly into Two-Dimensional AB2, AB5, and AB13 Superlattices,” Chem. Commun. 2006, 750-752. DOI: 10.1039/B515673D
29. C.-C. Chen, Y. Bisrat, Z. Luo, R.E. Schaak, C.-G. Chao, and D.C. Lagoudas, “Fabrication of Single Crystal Tin Nanowires by Hydraulic Pressure Injection,” Nanotechnology 2006, 17, 367-374. DOI: 10.1088/0957-4484/17/2/004
28. A.E. Henkes, J.C. Bauer, R.D. Johnson, A.K. Sra, R.E. Cable, and R.E. Schaak, “Low-Temperature Nanoparticle-Directed Solid State Synthesis of Ternary and Quaternary Transition Metal Oxides,” Chem. Mater. 2006, 18, 567-571. DOI: 10.1021/cm052190o
27. R.E. Cable and R.E. Schaak, “Low-Temperature Solution Synthesis of Nanocrystalline Binary Intermetallic Compounds using the Polyol Process,” Chem. Mater. 2005, 17, 6835-6841. DOI: 10.1021/cm0520113
26. Y. Vasquez, A.K. Sra, and R.E. Schaak, “One-Pot Synthesis of Hollow Superparamagnetic CoPt Nanospheres,” J. Am. Chem. Soc. 2005, 127, 12504-12505. DOI: 10.1021/ja054442s
25. B.M. Leonard, N.S.P. Bhuvanesh, and R.E. Schaak, “Low-Temperature Polyol Synthesis of AuCuSn2 and AuNiSn2: Using Solution Chemistry to Access Ternary Intermetallic Compounds as Nanocrystals,” J. Am. Chem. Soc. 2005, 127, 7326-7327. [Featured in Chemical & Engineering News (May 16, 2005) and Gold Bulletin (Vol. 38, #3, 2005)] DOI: 10.1021/ja051481v
24. R.E. Schaak, A.K. Sra, B.M. Leonard, R.E. Cable, J.C. Bauer, Y.-F. Han, J. Means, W. Teizer, Y. Vasquez, E.S. Funck, “Metallurgy in a Beaker: Nanoparticle Toolkit for the Rapid Low-Temperature Solution Synthesis of Functional Multimetallic Solid State Materials,” J. Am. Chem. Soc. 2005, 127, 3506-3515. [Featured in Chemical & Engineering News (May 16, 2005) and NanoTech Alert – Technical Insights (Frost & Sullivan, Feb 25, 2005)] DOI: 10.1021/ja043335f
23. T.D. Ewers, A.K. Sra, B.C. Norris, R.E. Cable, C.-H. Cheng, D.F. Shantz, and R.E. Schaak, “Spontaneous Hierarchical Assembly of Rhodium Nanoparticles into Spherical Aggregates and Superlattices,” Chem. Mater. 2005, 17, 514-520. DOI: 10.1021/cm0483792
22. A.K. Sra, T.D. Ewers, and R.E. Schaak, “Direct Solution Synthesis of Intermetallic AuCu and AuCu3 Nanocrystals and Nanowire Networks,” Chem. Mater. 2005, 17, 758-766. DOI: 10.1021/cm0484450
21. A.K. Sra and R.E. Schaak, “Synthesis of Atomically Ordered AuCu and AuCu3 Nanocrystals from Bimetallic Nanoparticle Precursors,” J. Am. Chem. Soc. 2004, 126, 6667-6672. DOI: 10.1021/ja031547r
20. N.M. Abrams and R.E. Schaak, “Self-Assembled Colloidal Crystals: Visualizing Atomic Crystal Chemistry using Microscopic Analogues of Inorganic Solids,” J. Chem. Educ. 2005, 82, 450-452. DOI: 10.1021/ed082p450
19. R.E. Schaak, R.E. Cable, B.M. Leonard, and B.C. Norris, “Colloidal Crystal Microarrays and Two-Dimensional Superstructures: A Versatile Approach for Patterned Surface Assembly,” Langmuir 2004, 20, 7293-7297. DOI: 10.1021/la049466b
18. R.J. Cavazos and R.E. Schaak, “Synthesis of the New Layered Oxides NaRbLnMO5 (Ln = La, Nd, Sm, Eu, Gd; M = Nb, Ta),” Mater. Res. Bull. 2004, 39, 1209-1214. DOI: 10.1016/j.materresbull.2004.04.014
17. R.E. Schaak and R.J. Cava, “Boron Substitution in Ternary Metal Phosphide Superconductors,” Mater. Res. Bull. 2004, 39, 1231-1235. DOI: 10.1016/j.materresbull.2004.04.011
16. J.W. Lynn, Q. Huang, C.M. Brown, V.L. Miller, M.L. Foo, R.E. Schaak, C.Y. Jones, E.A. Mackey, and R.J. Cava, “Structure and Dynamics of Superconducting NaxCoO2 Hydrate and Its Unhydrated Analog,” Phys. Rev. B 2003, 68, 214516.
15. B.G. Ueland, P. Schiffer, R.E. Schaak, M.L. Foo, V.L. Miller, and R.J. Cava, “Specific Heat Study of the Na0.3CoO2·1.3H2O Superconductor: Influence of the Complex Chemistry,” Physica C 2004, 402, 27-30. DOI: 10.1016/j.physc.2003.08.005
14. R.E. Schaak, T. Klimczuk, M.L. Foo, and R.J. Cava, “Superconductivity Phase Diagram of NaxCoO2·1.3H2O,” Nature 2003, 424, 527-529. DOI: 10.1038/nature01877
13. M.L. Foo, R.E. Schaak, V.L. Miller, T. Klimczuk, N.S. Rogado, Y. Wang, G.C. Lau, C. Craley, H.W. Zandbergen, N.P. Ong, and R.J. Cava, “Chemical Instability of the Cobalt Oxyhydrate Superconductor Under Ambient Conditions,” Solid State Commun. 2003, 127, 33-37. DOI: 10.1016/S0038-1098(03)00334-X
12. R.E. Schaak, M. Avdeev, W.-L. Lee, G. Lawes, H.W. Zandbergen, J.D. Jorgensen, N.P. Ong, A.P. Ramirez, and R.J. Cava, “Formation of Transition Metal Boride and Carbide Perovskites Related to Superconducting MgCNi3,” J. Solid State Chem. 2004, 177, 1244-1251. DOI: 10.1016/j.jssc.2003.10.032
11. R.E. Schaak and T.E. Mallouk, “Exfoliation of Layered Rutile and Perovskite Tungstates,” Chem. Commun. 2002, 706-707. DOI: 10.1039/B110220F
10. R.E. Schaak, D. Afzal, J.A. Schottenfeld, and T.E. Mallouk, “Na2Ln2Ti3-xMnxO10 (Ln = Sm, Eu, Gd, Dy; 0 ≤ x ≤ 1): A New Series of Ion-Exchangeable Layered Perovskites Containing B-Site Manganese,” Chem. Mater. 2002, 14, 442-448. DOI: 10.1021/cm010734q
9. R.E. Schaak and T.E. Mallouk, “Perovskites by Design: A Toolbox of Solid-State Reactions,” Chem. Mater. 2002, 14, 1455-1471. [Review article; featured on the cover, April 2002] DOI: 10.1021/cm010689m
8. R.E. Schaak and T.E. Mallouk, “KLnTiO4 (Ln = La, Nd, Sm, Eu, Gd, Dy): A New Series of Ruddlesden-Popper Phases Synthesized by Ion Exchange of HLnTiO4,” J. Solid State Chem. 2001, 161, 225-232. DOI: 10.1006/jssc.2001.9303
7. R.E. Schaak, E.N. Guidry, and T.E. Mallouk, “Converting a Layer Perovskite into a Non-Defective Higher-Order Homologue: Topochemical Synthesis of Eu2CaTi2O7,” Chem. Commun. 2001, 853-854. DOI: 10.1039/B009230B
6. R.E. Schaak and T.E. Mallouk, “Prying Apart Ruddlesden-Popper Phases: Exfoliation into Sheets and Nanotubes for Assembly of Perovskite Thin Films,” Chem. Mater. 2000, 12, 3427-3434. DOI: 10.1021/cm000495r
5. R.E. Schaak and T.E. Mallouk, “Self-Assembly of Tiled Perovskite Monolayer and Multilayer Thin Films,” Chem. Mater. 2000, 12, 2513-2516. DOI: 10.1021/cm0004073
4. R.E. Schaak and T.E. Mallouk, “Synthesis, Proton Exchange, and Topochemical Dehydration of New Ruddlesden-Popper Tantalates and Titanotantalates,” J. Solid State Chem. 2000, 155, 46-54. DOI: 10.1006/jssc.2000.8890
3. G.L. Egan, J.-S. Yu, C.H. Kim, S.J. Lee, R.E. Schaak, and T.E. Mallouk, “Nanoscale Metal Replicas of Colloidal Crystals,” Adv. Mater. 2000, 12, 1040-1042.
2. R.E. Schaak and T.E. Mallouk, “Topochemical Synthesis of Three-Dimensional Perovskites from Lamellar Precursors,” J. Am. Chem. Soc. 2000, 122, 2798-2803. DOI: 10.1021/ja993306i
1. R.E. Schaak, D.B. Dahlberg, and K.B. Miller, “The Electronic Nose: Studies on the Fundamental Response and Discriminative Power of Metal-Oxide Sensors,” Electronic Noses and Sensor Array Based Systems, Design and Applications, Proceedings of the 5th International Symposium of Olfaction and Electronic Nose, Ed. W. Jeffrey Hurst, 1999, Technomic Pub. Co., Inc.
Copyright © 2021 Schaak Lab, Penn State University – All Rights Reserved