Research focuses on cancer research including tumor cell adhesion and motility, tumor-endothelial interactions, and melanoma-induced signaling events. Past research has included developing a dual-micropipet technique to measure cell locomotion.
Current Studies
Tumor cell-induced Endothelial Barrier Disruption via VE-cadherin Disassembly and Cell Contractility
 In this study we are investigating the role of endothelial cell contractility and adherens junction disassembly in endothelial barrier breakdown during cell extravasation. Once regarded as a passive membrane, the endothelium is now recognized as an active player in multiple physiological functions and pathological conditions. In the context of cancer, cell extravasation across the endothelial barrier is necessary for the formation of secondary tumors. Previously we determined that interactions between melanoma and endothelial cells results in the disruption of the endothelial barrier. Moreover, we identified some of the signals used by melanoma cells to interact with endothelial cells such as cytokines and specific receptor-receptor interactions. We are currently using FRET biosensors, immunofluorescence and traction force microscopy techniques to de-couple and understand dynamic changes in endothelial cell contractility and adherens junction disassembly. The long-term objective of this project is to characterize the role of cytoskeletal tension and the molecular events leading to disassembly of adherens junctions in the process of cell extravasation. These studies will help to design better strategies to prevent the disruption of the endothelium.
In this study we are investigating the role of endothelial cell contractility and adherens junction disassembly in endothelial barrier breakdown during cell extravasation. Once regarded as a passive membrane, the endothelium is now recognized as an active player in multiple physiological functions and pathological conditions. In the context of cancer, cell extravasation across the endothelial barrier is necessary for the formation of secondary tumors. Previously we determined that interactions between melanoma and endothelial cells results in the disruption of the endothelial barrier. Moreover, we identified some of the signals used by melanoma cells to interact with endothelial cells such as cytokines and specific receptor-receptor interactions. We are currently using FRET biosensors, immunofluorescence and traction force microscopy techniques to de-couple and understand dynamic changes in endothelial cell contractility and adherens junction disassembly. The long-term objective of this project is to characterize the role of cytoskeletal tension and the molecular events leading to disassembly of adherens junctions in the process of cell extravasation. These studies will help to design better strategies to prevent the disruption of the endothelium.
In vitro Blood-Brain Barrier Models
 Brain cancer is a life-threatening disease due to its rapid development and the difficulty in its treatment. Even after an aggressive resection followed by concurrent or sequential radiation and chemotherapies, the median survival time of patients with the most common primary brain tumor, glioblastoma, is less than 15 months. Unfortunately, the delivery of drugs to the brain is extremely challenging due to the presence of the blood-brain barrier (BBB). To overcome this obstacle, we have been studying an immune cell-based nanoparticle delivery system using human immune cells as delivery vehicles. We have demonstrated its ability to transmigrate across the in vitro co-culture BBB model (figure) and effectively deliver therapeutic, biodegradable polymeric nanoparticles. This platform technology will allow us to attain a deeper understanding on the role of leukocytes in delivering drugs not only for brain cancer, but also for other neurological disorders that may cause a similar inflammatory response cascade that recruits leukocytes to the injury sites.
Brain cancer is a life-threatening disease due to its rapid development and the difficulty in its treatment. Even after an aggressive resection followed by concurrent or sequential radiation and chemotherapies, the median survival time of patients with the most common primary brain tumor, glioblastoma, is less than 15 months. Unfortunately, the delivery of drugs to the brain is extremely challenging due to the presence of the blood-brain barrier (BBB). To overcome this obstacle, we have been studying an immune cell-based nanoparticle delivery system using human immune cells as delivery vehicles. We have demonstrated its ability to transmigrate across the in vitro co-culture BBB model (figure) and effectively deliver therapeutic, biodegradable polymeric nanoparticles. This platform technology will allow us to attain a deeper understanding on the role of leukocytes in delivering drugs not only for brain cancer, but also for other neurological disorders that may cause a similar inflammatory response cascade that recruits leukocytes to the injury sites.
Mechanisms of Melanoma Tumor Cell Extravasation
 One of the principal requirements for cancer metastasis (cancer spread) is tumor cell adhesion and motility. Metastases from a primary tumor to secondary locations throughout the body are a major cause of cancer related deaths. In order to metastasize, tumor cells must undergo a series of steps. First they are shed into the blood stream (intravasation), survive while traveling through the blood vessels, and finally migrate again out of the vessels (extravasation) into a new location in the body. Finally they must proliferate.
One of the principal requirements for cancer metastasis (cancer spread) is tumor cell adhesion and motility. Metastases from a primary tumor to secondary locations throughout the body are a major cause of cancer related deaths. In order to metastasize, tumor cells must undergo a series of steps. First they are shed into the blood stream (intravasation), survive while traveling through the blood vessels, and finally migrate again out of the vessels (extravasation) into a new location in the body. Finally they must proliferate.
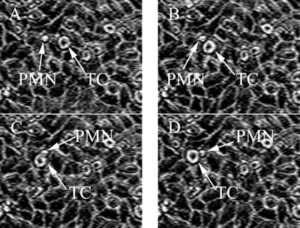 The interactions between cancer cells and the host immune system are of particular interest to our group. Innate immune system processes can potentially promote tumor progression through inflammation dependant mechanisms. White blood cells, neutrophils (PMN) in particular, are being studied to better understand how the host immune system affects cancer cell adhesion and subsequent migration in metastasis.
The interactions between cancer cells and the host immune system are of particular interest to our group. Innate immune system processes can potentially promote tumor progression through inflammation dependant mechanisms. White blood cells, neutrophils (PMN) in particular, are being studied to better understand how the host immune system affects cancer cell adhesion and subsequent migration in metastasis.
 This project studies the adhesion and extravasation stages of the tumor cell extravasation and metastatic process. Primarily, we are studying factors that promote a tumor cells’ ability to extravasate such as cellular dynamics, tumor-host interactions and relevant hemodynamic forces. Regulation of adhesion molecules (ICAM-1, integrins, etc.), cytokine/chemokine interactions and responses are a few studies currently being done. In addition, a migration assay has been developed to characterize tumor cell extravasation under shear flow conditions.
This project studies the adhesion and extravasation stages of the tumor cell extravasation and metastatic process. Primarily, we are studying factors that promote a tumor cells’ ability to extravasate such as cellular dynamics, tumor-host interactions and relevant hemodynamic forces. Regulation of adhesion molecules (ICAM-1, integrins, etc.), cytokine/chemokine interactions and responses are a few studies currently being done. In addition, a migration assay has been developed to characterize tumor cell extravasation under shear flow conditions.
Computational Fluid Dynamic (CFD) Modeling of Leukocyte Facilitated Tumor Cell Adhesion in a Shear Flow
 Previous work done in our lab has demonstrated that PMNs can facilitate tumor cell adhesion to the endothelium. Parallel plate flow chamber experiments have shown that under different flow conditions, melanoma cells and PMNs adhere to each other and to the endothelium with different efficiencies. These results have suggested a two-step adhesion mechanism in which first PMNs adhere to the endothelium, after which tumor cells adhere to those PMNs. The primary focus of this work is the development of a computational fluid dynamics, or CFD, model to explore what affect the fluid dynamics of the system have on the interactions between PMNs and melanoma cells in a shear flow.
Previous work done in our lab has demonstrated that PMNs can facilitate tumor cell adhesion to the endothelium. Parallel plate flow chamber experiments have shown that under different flow conditions, melanoma cells and PMNs adhere to each other and to the endothelium with different efficiencies. These results have suggested a two-step adhesion mechanism in which first PMNs adhere to the endothelium, after which tumor cells adhere to those PMNs. The primary focus of this work is the development of a computational fluid dynamics, or CFD, model to explore what affect the fluid dynamics of the system have on the interactions between PMNs and melanoma cells in a shear flow.
Tumor Cell Induced Endothelial Cell Junction Disassembly
 This study focuses on elucidating melanoma-induced signaling events leading to dissociation of endothelial cell junctional integrity. We have demonstrated that contact of melanoma cells to human umbilical vein endothelial cells (HUVEC) triggered rapid endothelial [Ca2+]i response. In addition, alternation of endothelial adherens junctions following contact of melanoma cell was also evidenced by the changes in tyrosine phosphorylation of vascular endothelial VE-cadherin. Melanoma contact induced a time-dependent increase in tyrosine phosphorylation. Tyrosine phosphorylation reached a maximum at 45 min following the introduction of melanoma cells and diminished thereafter. The immunological staining patterns of VE-cadherin are strongly peripheral and junctional structures are intact prior to melanoma contact (t = 0). Upon melanoma contact with endothelium, highly localized dissociation of these junctions occurs (t = 45 min). In addition, signaling molecules besides Ca2+ and receptors involved in controlling the downstream junctional regulation following contacts with melanoma cells are being studied.
This study focuses on elucidating melanoma-induced signaling events leading to dissociation of endothelial cell junctional integrity. We have demonstrated that contact of melanoma cells to human umbilical vein endothelial cells (HUVEC) triggered rapid endothelial [Ca2+]i response. In addition, alternation of endothelial adherens junctions following contact of melanoma cell was also evidenced by the changes in tyrosine phosphorylation of vascular endothelial VE-cadherin. Melanoma contact induced a time-dependent increase in tyrosine phosphorylation. Tyrosine phosphorylation reached a maximum at 45 min following the introduction of melanoma cells and diminished thereafter. The immunological staining patterns of VE-cadherin are strongly peripheral and junctional structures are intact prior to melanoma contact (t = 0). Upon melanoma contact with endothelium, highly localized dissociation of these junctions occurs (t = 45 min). In addition, signaling molecules besides Ca2+ and receptors involved in controlling the downstream junctional regulation following contacts with melanoma cells are being studied.
Previous Studies
Melanoma Cell Migration to Type IV Collagen Requires Activation of NF-kB
Chemotaxis is the consequence of environmental factors engaging their receptors to initiate signaling cascades. However, the biochemical mechanisms controlling this phenomenon are not clear. We employed an in vitro modified Boyden 48-well chemotaxis migration system to characterize the role of signal transducers in type IV collagen-induced A2058 human melanoma cell migration. Using specific pharmacological inhibitors and a series of dominant-negative and constitutively active signaling proteins, we show that Ras and Rac GTPases, PI3-K, and PKC participate in cell migration mediated by β1 integrins. Collagen also induces a time-dependent degradation of IkB-a and an increase in nuclear translocation of NF-kB which is dependent on PKC pathway. More importantly, collagen-stimulated melanoma cell migration directly correlated with an increase in NF-kB transactivation. Furthermore, CIV induced an increase in β1 integrin mRNA levels. Specific NF-kB inhibitors Helenalin and SN-50 inhibited melanoma cell migration to collagen, indicating a novel requirement for NF-kB transactivation in cell chemotaxis mediated by β1 integrin signals. These results describe signal transduction events that are initiated by type IV collagen through β1 integrins and demonstrate an important role for NF-kB in regulating melanoma chemotaxis.

Cell Deformation and Adhesion Studies
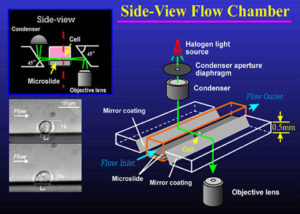 Observing microscopic specimens is often useful in studies of cellular interaction with a vascular wall. We have developed an in vitro side-view flow chamber that permits observations from the side of the cell’s contact with various adhesive surfaces under dynamic flow conditions. This side-view flow chamber consists of two precision rectangular glass tubes called microslides. A smaller microslide is inserted into a larger one to create a flow channel with a flat surface on which either cultured vascular endothelium can be grown or purified adhesion molecules can be coated. Two optical prisms with a 45 degree angle chromium-coated surface are used along the flow channel to generate light illumination and observation pathways. The side-view images of cell-substrate contact can be obtained using a light microscope. This design allows us not only to measure the effects of flow on cell-surface adhesion strength, but also to have close observation of cell deformation and adhesive contact to various surfaces in shear flow. Using this side-view flow assay, the mechanics of white blood cell deformation and adhesion to various endothelia cell adhesion molecules in dynamic shear flow has been investigated.
Observing microscopic specimens is often useful in studies of cellular interaction with a vascular wall. We have developed an in vitro side-view flow chamber that permits observations from the side of the cell’s contact with various adhesive surfaces under dynamic flow conditions. This side-view flow chamber consists of two precision rectangular glass tubes called microslides. A smaller microslide is inserted into a larger one to create a flow channel with a flat surface on which either cultured vascular endothelium can be grown or purified adhesion molecules can be coated. Two optical prisms with a 45 degree angle chromium-coated surface are used along the flow channel to generate light illumination and observation pathways. The side-view images of cell-substrate contact can be obtained using a light microscope. This design allows us not only to measure the effects of flow on cell-surface adhesion strength, but also to have close observation of cell deformation and adhesive contact to various surfaces in shear flow. Using this side-view flow assay, the mechanics of white blood cell deformation and adhesion to various endothelia cell adhesion molecules in dynamic shear flow has been investigated.
MMP Studies
This study aims to discover the mechanisms of tumor-endothelial interactions that aid the melanoma cell in invasion. Matrix Metalloproteinase (MMPs), in particular, have been implicated in cancer angiogenesis, invasion, migration and metastasis. In this study, we focus on the gelatinases (MMP2, 9) and collagenase (MMP1). To study the role that MMPs play in melanoma metastasis, melanoma invasion was studied using a novel in-vitro chemotaxis invasion chamber. Endothelial cells were grown on a layer of Matrigel that was coated on a filter in an invasion assay to simulate the in vivo tumor extravasation and invasion process through the blood vessel wall. There is strong evidence to suggest that IL-8 is responsible for increased expression of MMP2 into the supernatant. MMP2, a gelatinase, has been implicated in both metastasis and angiogenesis in cancer and this allows us to understand more of the tumor microenvironment in order to effectively apply therapy.
Lipid Mediated Signaling in Tumor Cell Activation
 This study focuses on effects of extracellular lipid groups on tumor cell pseudopodial protrusion and cell migration in vitro. We are interested in understanding the interaction between extracellular ligands (extracellular matrix protein molecules) with cell receptors, and how extracellular bioactive lipids contribute to such signaling mechanisms. Together with the initial signaling mechanism, we are characterizing the subsequent cell activation via Ca2+ secondary messenger mediated pathway and how that affects the cellular cytoskeleton reorganization. Glass micropipette assay system (Figure A~C) enables us to focally apply the chemotactic stimulant solution onto a specific region of a suspended cell; a cell protruding pseudopods can be observed and characterized in real-time using brightfield or fluorescence microscopy. Adhered cells spreading and migrating toward chemotactic stimulation can also be assayed in real-time using our system (Figure D).
This study focuses on effects of extracellular lipid groups on tumor cell pseudopodial protrusion and cell migration in vitro. We are interested in understanding the interaction between extracellular ligands (extracellular matrix protein molecules) with cell receptors, and how extracellular bioactive lipids contribute to such signaling mechanisms. Together with the initial signaling mechanism, we are characterizing the subsequent cell activation via Ca2+ secondary messenger mediated pathway and how that affects the cellular cytoskeleton reorganization. Glass micropipette assay system (Figure A~C) enables us to focally apply the chemotactic stimulant solution onto a specific region of a suspended cell; a cell protruding pseudopods can be observed and characterized in real-time using brightfield or fluorescence microscopy. Adhered cells spreading and migrating toward chemotactic stimulation can also be assayed in real-time using our system (Figure D).
Cell Motility and Locomotion Studies
 We have developed a dual-micropipet technique to measure cell locomotion. the objective of this work was to characterize tumor cell locomotion in response to chemotactic stimulation. The assay involves two micropipets. An individual tumor cell (A2058 human melanoma cell is shown) was retained, without pressure gradient, in a pipette of approximately 14 microns i.d. A solution of type IV collagen (one of the important extracellular matrix proteins), chosen as the chemotactic source, was placed in another pipette (~10 microns o.d.) with zero pressure at the pipette tip. The smaller pipette was then inserted into the larger one containing the melanoma cell. The initial chemoattractant concentration and the distance between the tip of the small pipette and the cell surface provided a gradient for tumor cell locomotion toward stimulation. This novel assay provides a direct measure of cell movement: cyclic pseudopod protrusion and subsequent cell locomotion. The influences of different adhesion substrates on cell locomotion were also studied.
We have developed a dual-micropipet technique to measure cell locomotion. the objective of this work was to characterize tumor cell locomotion in response to chemotactic stimulation. The assay involves two micropipets. An individual tumor cell (A2058 human melanoma cell is shown) was retained, without pressure gradient, in a pipette of approximately 14 microns i.d. A solution of type IV collagen (one of the important extracellular matrix proteins), chosen as the chemotactic source, was placed in another pipette (~10 microns o.d.) with zero pressure at the pipette tip. The smaller pipette was then inserted into the larger one containing the melanoma cell. The initial chemoattractant concentration and the distance between the tip of the small pipette and the cell surface provided a gradient for tumor cell locomotion toward stimulation. This novel assay provides a direct measure of cell movement: cyclic pseudopod protrusion and subsequent cell locomotion. The influences of different adhesion substrates on cell locomotion were also studied.