The Gill Lab – Understanding Calcium Signal Transduction
Calcium is one of the most fundamental signaling agents in all animal cells. Cells have evolved to precisely control Ca2+ in the cytoplasm at levels that are 10,000-fold lower than outside cells. This is accomplished by Ca2+ pumps in the plasma membrane (PM) and endoplasmic reticulum (ER). We study the signals of Ca2+ which occur as a result of control of specific channels in the PM and ER membrane which allow Ca2+ to flow into the cytosol. A slight elevation in the resting cytosolic Ca2+ level is enough to trigger rapid cellular responses such as contraction, secretion or changes in the function of key metabolic enzymes. More sustained Ca2+ signals mediate crucial longer term responses including cell growth, cell division, and cell death (apoptosis). Our lab studies signal transduction, meaning that we study how cells transduce external signals into Ca2+ signals. Cells sense many different external signals through specific receptors for chemical agents such as growth factors, neurotransmitters, and hormones, as well as receptors for temperature, pressure, stretch, sound, and light. The cell converts the message received by receptors into Ca2+ signals by precisely controlling the opening of Ca2+ channels. We use a combination of molecular biology, biochemistry, cell biology, and single cell physiological approaches to understand how the Ca2+ channels are controlled. We use molecular biology to mutate the channel proteins, create expression vectors, and to modify channel expression using gene silencing approaches. We follow real-time Ca2+ signals in cells using sophisticated single cell ratiometric fluorescence imaging technology. And we measure the precise biophysical properties of channels using state-of the-art electrophysiological methods. The work centers on the analysis of several distinct types of membrane channels including members of the now widely recognized TRP family of channel proteins involved in transducing a remarkable array of external signals. More recently, we have focused on understanding the mechanisms by which STIM and Orai proteins are involved in the controlling Ca2+ signals. Our work draws together molecular and cellular approaches to understand the basic function and physiological role of these channels which are critical to mediating essential cellular responses.
Why are Calcium Signals So Important to Study?
From fertilization to cell death, Ca2+ signals instigate and coordinate critical cell functions. The remarkable spatial and temporal precision of Ca2+ signals reflects the function of highly coordinated Ca2+ sensors and Ca2+ channels. The recently identified and ubiquitous STIM-Orai signaling pathway, is mediated through the dynamic interactions between STIM Ca2+ sensor proteins in the ER, and the family of highly Ca2+-selective Orai channels in the PM. The two proteins physically interact in ER-PM junctions mediating “store-operated” Ca2+ entry (SOCE), crucial in controlling gene expression, secretion, motility and growth of all cell types. Coupling between STIM and Orai proteins is an evolutionarily conserved, master control-circuit for Ca2+ signaling. Ca2+ stored in ER is critical to both ER function and Ca2+ signal generation. STIMs constantly monitor ER Ca2+ and couple with Orai channels to ensure Ca2+ homeostasis. SOCE is critical in generating two distinct types of Ca2+ signals: spatially discrete local Ca2+ entry signals at the ER-PM junction, and global Ca2+ oscillations that travel across the cytosol and penetrate the nucleus (Fig.1). STIMs sense depleted ER Ca2+, unfold, and move into ER-PM junctions where they trap and open Orai channels to allow Ca2+ entry. STIM1 and STIM2 have different ER sensing thresholds and distinct Orai channel coupling efficacy. Although receptor-induced InsP3 triggers the oscillatory Ca2+ release, SOCE-mediated store refilling supplies the Ca2+ necessary for generating oscillations, and hence profoundly controls the amplitude, frequency and duration of Ca2+ oscillations. In addition, the highly localized Ca2+ entry signals at STIM-Orai junctions play a critical role in activating calcineurin, dephosphorylating cytosolic NFAT and triggering its nuclear localization to control transcription. This subtle local Ca2+ entry signal is very different to the large global Ca2+ oscillations, but they are highly coordinated and interdependent. For example, the Ca2+ entry and release signals differentially impinge on NFAT function and c-fos immediate early gene expression – local SOCE signals dephosphorylate and prompt nuclear entry of NFAT1 and NFAT4, whereas global nuclear-penetrating Ca2+ oscillations inhibit rephosphorylation and delay nuclear export of NFAT4 The amplitude of Ca2+ oscillations also controls NF-kB activation. STIM proteins are also broader stress-sensors, responding to redox, hypoxia, pH, and temperature changes to control Orai channels. STIM proteins also control other target channels including TRPC and voltage-operated Ca2+ channels.
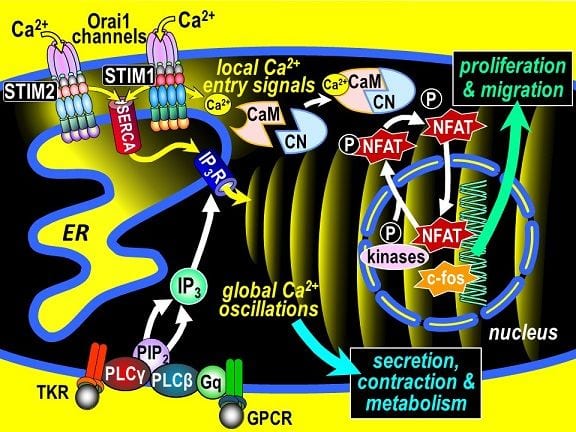 Store-Operated Calcium Entry controls both local and global Ca2+ signals. The two STIM proteins, STIM1 and STIM2, are sensors of Ca2+ levels in the endoplasmic reticulum (ER). Store-depletion by inositol trisphosphate receptor Ca2+ release channels (IP3Rs) activates STIM proteins and they become trapped within ER-PM junctions. STIM2 is more sensitive to store depletion than STIM1. In these junctions, the STIM proteins tether and open Orai channels. Orai channels (Orai1, Orai2, and Orai3), are highly selective Ca2+ entry channels. Orai channel opening mediates local Ca2+ entry signals that are crucial in cells. For example, the Ca2+ entering through Orai channels controls activation of the major transcription factor, NFAT. The Ca2+ activates CaM/CN leading to dephosphorylation and nuclear localization of NFAT, where it regulates genes controlling growth, migration, and division of cells. The store-operated Ca2+ entry is also crucial for sustaining global Ca2+ oscillations, Ca2+ signals controlling a huge spectrum of vital cellular events including secretion, contraction, and metabolism. Altered expression and mutation of STIM and Orai proteins have been implicated in a great many human disorders including severe combined immunodeficiency, muscular hypotonia, autoimmunity, skin dysplasia, and exocrine defects. Dysregulation of STIM/Orai expression is closely linked to cardiovascular and airway remodeling, neurodegenerative disorders, altered immunity, and cancer.
Store-Operated Calcium Entry controls both local and global Ca2+ signals. The two STIM proteins, STIM1 and STIM2, are sensors of Ca2+ levels in the endoplasmic reticulum (ER). Store-depletion by inositol trisphosphate receptor Ca2+ release channels (IP3Rs) activates STIM proteins and they become trapped within ER-PM junctions. STIM2 is more sensitive to store depletion than STIM1. In these junctions, the STIM proteins tether and open Orai channels. Orai channels (Orai1, Orai2, and Orai3), are highly selective Ca2+ entry channels. Orai channel opening mediates local Ca2+ entry signals that are crucial in cells. For example, the Ca2+ entering through Orai channels controls activation of the major transcription factor, NFAT. The Ca2+ activates CaM/CN leading to dephosphorylation and nuclear localization of NFAT, where it regulates genes controlling growth, migration, and division of cells. The store-operated Ca2+ entry is also crucial for sustaining global Ca2+ oscillations, Ca2+ signals controlling a huge spectrum of vital cellular events including secretion, contraction, and metabolism. Altered expression and mutation of STIM and Orai proteins have been implicated in a great many human disorders including severe combined immunodeficiency, muscular hypotonia, autoimmunity, skin dysplasia, and exocrine defects. Dysregulation of STIM/Orai expression is closely linked to cardiovascular and airway remodeling, neurodegenerative disorders, altered immunity, and cancer.
