Aug 2018: Graduate Student Retreat at Hershey
Best Posters by Rob and Xiangyu! Guess who won the Prize!!
JBC – Editor’s Pick August 2018!! Congratulations Xiangyu!
Pore properties of Orai1 calcium channel dimers and their activation by the STIM1 ER calcium sensor
Xiangyu Cai, Robert M. Nwokonko, Natalia A. Loktionova, Raz Abdulqadir, James H. Baraniak, Jr., Youjun Wang, Mohamed Trebak,
Yandong Zhou, and Donald L. Gill
A Real Breakthrough: Congratulations Yandong and Robert!!
Cross-Linking Paper in PNAS April 2018
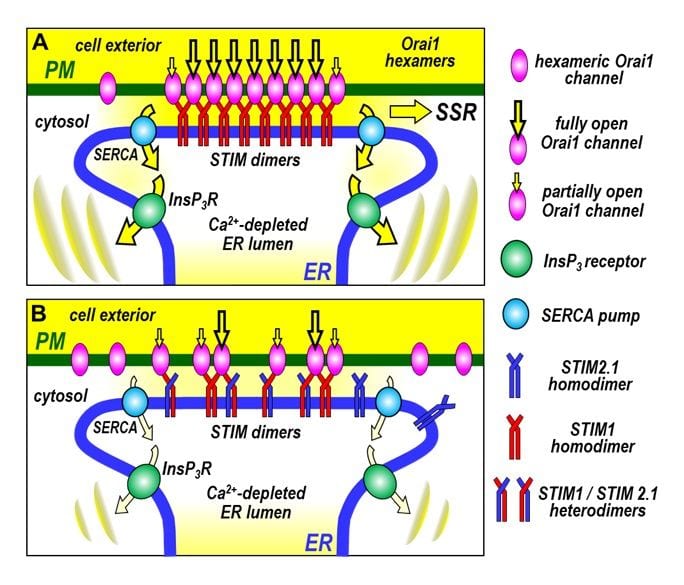
Model for the cross-linking of Orai channels by STIM proteins and its physiological role in the generation of Ca2+ signals. (A) When STIM1 is the major STIM protein isoform present, the homodimers of STIM1 moving into ER-PM junctions are able to cross-link hexameric Orai1 channels, increasing the efficacy, rate and density of Orai1 channel activation in junctions. This results in a maximal number of activated Orai1 channels mediating store-operated Ca2+ entry (SOCE) at the ER-PM junction. The larger SOCE allows efficient Ca2+ store-refilling via sarco/endoplasmic reticulum Ca2+ ATPase (SERCA) Ca2+ pumps, which sustains the generation of oscillatory Ca2+ release signals mediated by inositol trisphosphate receptors (InsP3R). The enhanced SOCE may also increases downstream “SOCE signature responses” (SSR), for example, activation of calcineurin and NFAT. (B) When cells express the splice variant, STIM2.1 together with STIM1, heterodimers of STIM1 and STIM2.1 move into junctions, but can only undergo monomeric interactions with Orai1 channels. They are unable to cross-link Orai1 channels and hence prevent clustering. This results in lowered efficacy, rate and density of Orai1 channel activation. The decreased SOCE results in lowered store-refilling, and the inability of cells to sustain the generation of oscillatory Ca2+ release signals. The decreased SOCE may also reduce SSR. Thus, STIM2.1 may act as a powerful regulator of SOCE. Note that STIM2.1 homodimers play no role; they cannot bind Orai1, nor can they directly influence cross-linking.