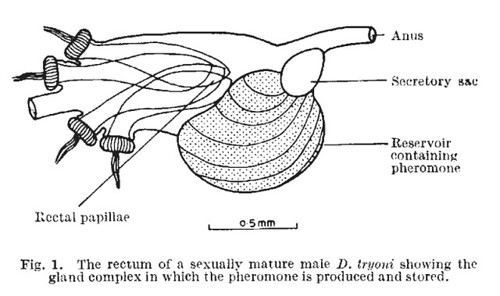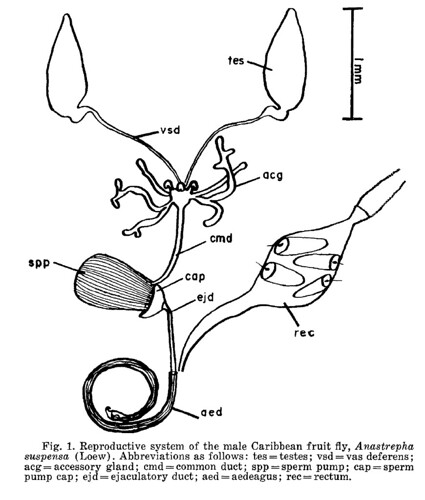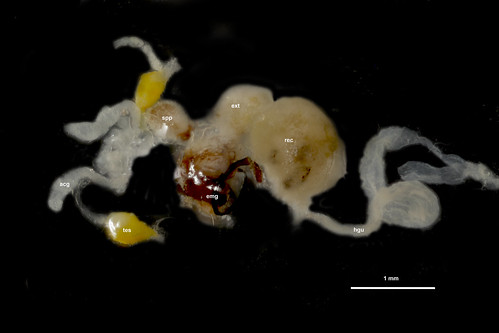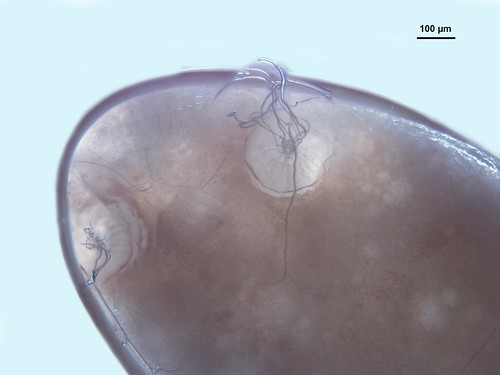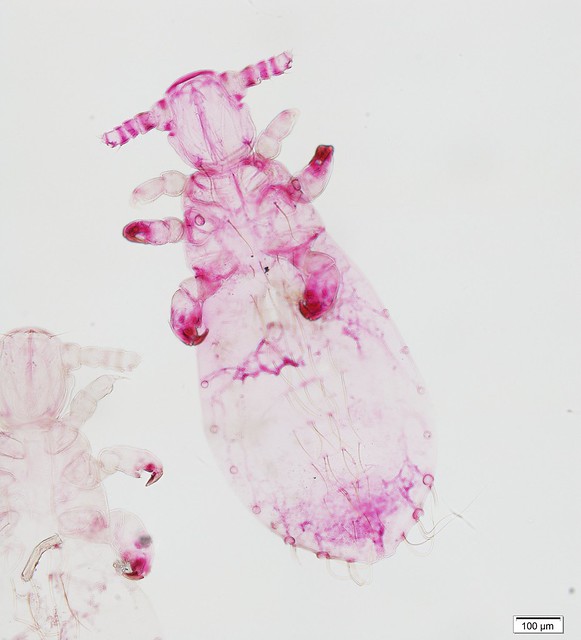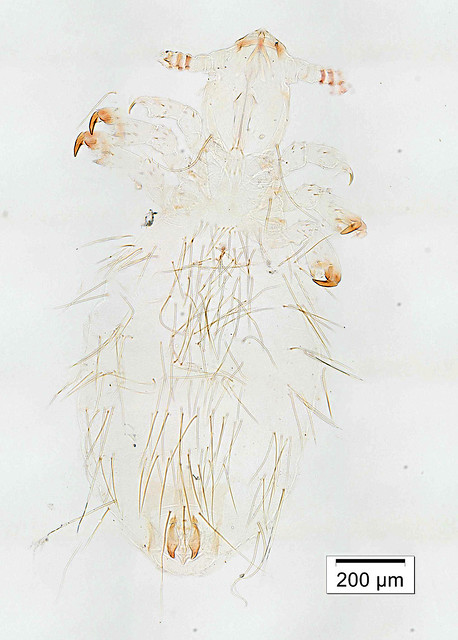
This week’s Friday Night Lice mystery louse parasitizes a host from an order of mammals we haven’t yet covered in this series. So, it’s not found on rodents, carnivores, pinnipeds, primates … Can you guess what it is? It was collected in Nepal in the late 1960s. Offer your guesses as comments below! We’ll post the answer on Monday.