My previous research at Penn State covered the core areas of colloids, polymers and soft nanomaterials with the focus on molecular self-assembly and the resulting system of soft nanocolloids such as micelles, vesicles, microemulsions, polymer-micelle complexes, block copolymer micelles, etc. My research was directed towards the complementary goals of developing predictive molecular theories of complex systems (involving surfactants, block copolymers, colloids, proteins and enzymes) and exploiting such complex systems for practical applications relevant to industry.
My current work at NSRDEC is focused on applying my expertise in colloids, polymers and nanomaterials science with special emphasis on the self-assembly process to Warfighter-related fundamental and early applied research problems. Over the last ten years, my lab has been involved in the following research efforts:
Stable Carbon Nanotube Dispersions using Self-Assembly

Carbon nanotubes (CNTs) exist as aggregates limiting their practical use. Current approaches to disperse CNT either reduce CNT functionalities or impart only kinetic stability. Our goal is to develop thermodynamically stable CNT dispersions by taking advantage of the self-assembly properties of block copolymers in solution and their ability to solubilize carbon nanotubes. The stability imparted to nanotube dispersions will allow them to be applied as coatings on textiles for providing electrical and mechanical properties to soldier uniform materials. This work also revealed that it would be possible to separate nanotubes by their size using the approach developed here, which is of interest to many applications of nanotechnology. (Published in J Chem Phys 2009)
Adhesive, Barrier Material for Closure Application
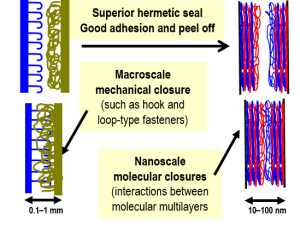
There is a need for closure systems for use in personal protective equipment of Warfighters to provide hermetic sealing against any chemical vapor threats in the field. Our goals is to develop a non-mechanical, molecular adhesion-based closure system based on nanoscale polymer multilayers to provide such hermetic sealing, sufficient adhesive strength, and peel-off ease. We developed a polymer multilayer system by self-assembly of oppositely charged polyelectrolytes, as a replacement for the conventional Velcro closures used in soldier protective clothing and equipment. The nanoclosures were shown to provide significantly enhanced barrier to gas permeation compared to Velcro but did not possess sufficient mechanical strength. (Published in ACS Applied Materials and Interfaces, 2012)
Mechanism of Action of Antimicrobial Peptides

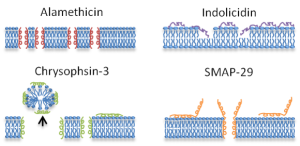
We are interested in differentiating the action of different antimicrobial peptides on cell membranes by identifying the molecular scale mechanisms involved (adsorption, membrane insertion, pore formation, membrane lysis). This is being done by combining QCM-D experiments on supported lipid bilayers used as model membranes with analytical theory of peptide-lipid bilayer interactions. This study of nanoscale interactions between peptides and lipid bilayers self-assembled as membranes helped identify the mechanisms by which the peptides break down walls of cell membranes, depending on the peptide molecular properties. This work offers the potential to identify appropriate peptide molecular structures that can be covalently bound to textile surfaces in order to provide antimicrobial protection to the soldier. (Published in J Phys Chem B 2011 and Colloids Surf B 2014)
Nanoparticle Interactions with Membranes

Knowledge of nanoparticle (NP) – membrane interactions is important to advances in nanomedicine as well as for determining the safety of NPs to humans and the ecosystem. This study focuses on a unique mechanism of cytotoxicity, cell membrane destabilization, which is principally dependent on the nanoparticle nature of the material rather than on its molecular properties. We investigated the interactions of gold NPs with supported lipid bilayer using QCM-D, both in the absence and presence of polymethacrylic acid (PMAA), used to simulate the natural organic matter in the environment. We have proposed a simple model to show that the competition between the particle-bilayer adhesion energy, the bilayer bending energy, and the interfacial energy at bilayer defect edges allows the larger NPs which become more adhesive because of the polymer adsorption, to be engulfed by the bilayer and leave the crystal surface, causing large mass loss and membrane disruption. The QCM-D measurements thus offer direct evidence that even if NPs are intrinsically not cytotoxic, they can become cytotoxic in the presence of environmental organic matter which modulates the adhesive interactions between the nanoparticle and the membrane. (Published in Biophys Chem 2015)
Nanoparticle Mediated Stable Polymer Blends

Morphology dictates the properties of polymer blends and composites. We propose a strategy to control morphologies of polymer blends by generating thermodynamically stable polymer-polymer microemulsions with block copolymers as “surfactants” and nanoparticles as “co-surfactants”. This effort at creation of a polymer-polymer-nanoparticle composite was driven by the need to achieve good dispersion and stability of the nanoparticle-polymer system. Potential application of such materials is to achieve vastly improved mechanical performance and /or electrical properties for use in soldier materials and reduce the risk of property loss due to thermodynamic instability during material processing steps. We also demonstrated that polymer-nanoparticle systems can be prepared for potential application as thinner and more dexterous gloves, without compromising on the protective barrier properties.
Computational chemistry approaches to predicting permeation of chemical agents through Warfighter protective clothing
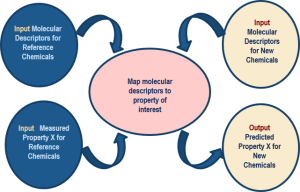
Quantitative knowledge of chemical warfare agents (CWA) permeation through protective barrier materials is difficult to obtain due to their extreme toxicity and lethality. Our goal is to develop purely computational methods to predict CWA permeation without having to perform experiments using CWAs. This effort allowed us to bring the computational chemistry techniques widely practiced in drug discovery to the study of chemical warfare agents. It allowed us to predict the permeation of chemical agents through protective barrier materials using data driven computational chemistry calculations.
Molecular Similarity Based Simulant Search

In this research, we develop a systematic and quantitative methodology to identify or design simulants for chemical warfare agents and emerging future threats, using molecular similarity analysis and exploiting advances in drug discovery methods developed in the pharmaceutical industry. Potentially, the simulant selection/design can also be optimized with its toxicity behavior. (Published in J Hazard Mat 2011)
Multifunctional polymer nanoparticles by self-assembly

Multifunctional polymer nanoparticles of variable sizes and functionalities can be produced starting from the same block copolymer by end functionalizing the block copolymer with desired functionalities, controlling the choice of solvent and incorporating desired proportion of different functional groups, by self-assembly. This effort is allowing us to create polymer nanoparticles of controlled size and composition with multiple functionalities for pathogen detection, antimicrobial action, cell penetration and MRI imaging capabilities. The multifunctional nanoparticles can be potentially applied as coatings for soldier uniform materials or can be covalently attached to surfaces to create intelligent surfaces or can be used for biomedicine (Published in RSC Advances, 2014).
Aqueous based production of electrospun polymer nanofibers

Our goal is to develop an environmentally conducive green solvent method for electrospinning to produce novel polymer nanofiber materials. We have demonstrated the use of polymer-micelle complex as an aid to electrospin in aqueous solutions. The novel process uses the self-assembled polymer-surfactant micelle system to control the conductivity, viscosity and surface tension of the polymer solution being electrospun. We have electrospun nanofibers incorporating carbon nanotubes and proteins and have characterized the surface through AFM adhesion profiles. This process development will allow polymer nanofiber production and the use of functionalized nanofibers as coatings on uniforms for sensing and protection and also for applications such as filters. (Published in J Phys Chem C 2007; U.S. Patent Number 9,175,422 B2 awarded on 3 Nov 2015; ACS Applied Materials and Interfaces, 2014)
Integrated Systems Approach to Optimization of Point-of-Use (PoU) Decontaminant Formulation, Delivery and Application

Decontamination formulation development is traditionally divorced from how it is applied. The simultaneous optimization of the decontaminant formulation, delivery and application methods will make the integrated decontamination process more effective at the point-of-use with minimal logistic needs. This research is aimed at generating decontaminant formulations, delivery and application methods that are optimized for the point of use depending on the type of chemical agent threat encountered, predominant material types that are contaminated and the logistics such as the quality and amount of water available. We have custom designed and built spray systems for conducting engineering optimization experiments.
Design of nanostructured enzyme-micelle conjugate by self-assembly for decontamination of sensitive surfaces such as electronic components on soldier equipment or aircraft interiors

Our goal is to develop a novel decontaminant based on a conjugate of block copolymer with the OPH enzyme. This will allow integration of multiple functionalities within the formulation including the displacement of agents from surfaces, sequestering agents within micelles, bringing agent to the proximity of enzyme active site, improving the stability of enzyme and its shelf life. In this research, we demonstrated enhanced stability and biological activity of organophosphate hydrolase, one of the most important organophosphate agent inactivating enzymes, under a variety of stressors including high temperatures, presence of organic solvents, lyophilization and freeze thawing (for the storage of enzymes), by creating aggregates of covalent polymer-enzyme conjugates. (Published in Biomacromolecules, 2014)
Inactivation of Bacterial Spores

Our goal is to understand the mechanisms of bacterial spore inactivation/killing using liquid based spore inactivation experiments and nanoscale surface characterization with Atomic Force Microscopy. Also single spore characterization is done using suspended microchannel resonator (SMR) experiments. The results would aid in the development of sporicidal treatments on surfaces.
An Autonomous, Adaptive, Responsive Second Skin Based on an Organohydrogel Platform

This research effort is aimed at generating next generation protective barrier material resembling second skin using combinations of nanomaterials, enzymes, and polymer gels to create an autonomously functioning active, responsive material system. The second skin is constructed on a non-woven textile substrate. The polydiactylene-pralidoxime conjugate micelles bind OP agents selectively and undergo color change. The nucleophilic PAAm-APy polymer promotes hydrolysis of OP agents (G and VX) to create acidic products that cause gel swelling. In brominated state, the polymer inactivates anthrax spores and also degrades mustard. The porous silica nanoparticle impregnated with 8-hydroxy quinone as catalyst an adsorb and degrade HD generating non-toxic acidic products. The acidic products cause gel swelling and also offer a way to sense HD. Within the PEGDA-PAA-silicone organohydrogel (OHG), hydrolysis of HD causes a pH change resulting in gel swelling. The pH change is also colorimetrically monitored. The droplets within the OHG also aggregate to create a permeation barrier. The PEGDA-PAA polymer in the OHG has oximes attached as pendent groups. It also has disulfide cross-links. VX reacts with oxime to generate a thiolate which disrupts the disulfide cross-link by thiol-disulfide exchange reaction, causing gel to swell. Reaction product of VX with oxime is non-toxic. The carbon fiber provides additional protection if used.
“Molecules” from Nanoparticle “Atoms”
“Molecules” composed of various kinds of nanoparticles as ‘giant atoms’ offer the potential for new materials, with remarkably enhanced properties and functionalities. In this work, we describe a robust substrate-based step-by-step assembly strategy to create such molecules by tuning their interactions to enable controlled covalent assembly, while avoiding aggregation. Plasmonic gold and silver nanoparticles anchored on a substrate was used for the controlled formation of stable oligomeric molecules, bridged by ‘linkers”. The nature of the constituent nanomaterials, their surface chemistry (type of capping ligands, ligand packing), choice of linkers and other experimental parameters influenced the resulting nanoparticle assembly on the substrate. Following the attachment of an anchor nanoparticle on the substrate, we investigated methods to selectively remove/deactivate the excess linkers present on the substrate, to prevent nonselective adsorption of the second nanoparticles on the substrate but instead promote their attachment to the anchor nanoparticles to build the molecule. An array of homodimers (same metal and shape and size), heterodimers (different metal or shape or size) and satellite nanoparticles (consisting of a core and ‘satellites’ surrounding them) were successfully synthesized using this strategy and characterized using electron microscopic techniques. By tailoring the contact times of the substrate in the nanoparticle solutions as well as by varying the concentration of the nanoparticle solution, the number of satellite nanoparticles on the ‘core-satellite’ structures can be tuned. These colloidal molecules built on the substrate can be utilized for applications including sensing, catalysis, and SERS.
